UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, DC 20549
FORM
CURRENT REPORT
Pursuant to Section 13 or 15(d)
of the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported):
(Exact name of registrant as specified in its charter)
| (State or other jurisdiction of incorporation or organization) |
(Commission File Number) | (I.R.S. Employer Identification No.) |
|
|
||
| (Address of principal executive offices) | (Zip Code) |
Registrant’s telephone number, including
area code: (
Not Applicable
(Former name or former address, if changed since last report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions (see General Instruction A.2. below):
| Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) | |
| Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) | |
| Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) | |
| Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
| Title of Each Class | Trading Symbol | Name of Exchange on Which Registered | ||
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company
If an emerging growth company, indicate by check mark if the registrant
has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant
to Section 13(a) of the Exchange Act.
| Item 2.02 | Results of Operations and Financial Condition |
On March 19, 2025, Century Therapeutics, Inc. (the “Company”) issued a press release announcing its financial results for the year ended December 31, 2024. A copy of the press release is furnished as Exhibit 99.1 to this Current Report on Form 8-K and is incorporated herein by reference.
The information contained in this Item 2.02 (including Exhibit 99.1) is being furnished and shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that section and shall not be deemed to be incorporated by reference in any filing under the Securities Act of 1933, as amended, or the Exchange Act, except as shall be expressly set forth by specific reference in such filing.
| Item 7.01 | Regulation FD Disclosure |
On March 19, 2025, the Company updated information reflected in a slide presentation, which is attached as Exhibit 99.2 to this Current Report on Form 8-K and is incorporated herein by reference. Representatives of the Company will use the updated presentation in various meetings with investors from time to time.
The information contained in this Item 7.01 (including Exhibit 99.2) is being furnished and shall not be deemed “filed” for purposes of Section 18 of the Exchange Act, or otherwise subject to the liabilities of that section and shall not be deemed incorporated by reference in any filing under the Securities Act or the Exchange Act, except as shall be expressly set forth by specific reference in such filing.
| Item 9.01 | Financial Statements and Exhibits |
(d) Exhibits
| Exhibit No. |
Document | |
| 99.1 | Press Release of Century Therapeutics, Inc., dated March 19, 2025 | |
| 99.2 | Investor Presentation of Century Therapeutics, Inc., dated March 19, 2025 | |
| 104 | Cover Page Interactive Data File (embedded within the Inline XBRL document) |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| CENTURY THERAPEUTICS, INC. | ||
| By: | /s/ Brent Pfeiffenberger, Pharm.D. | |
| Name: | Brent Pfeiffenberger, Pharm.D. | |
| Title: | President and Chief Executive Officer | |
| Date: March 19, 2025 | ||
Exhibit 99.1
Century Therapeutics Reports Full Year 2024 Financial Results and Provides Business Update
| · | Preclinical pipeline re-prioritization to focus on four potentially transformative programs to advance toward clinic, led by CNTY-308 in B-cell mediated autoimmune diseases and malignancies |
| · | New concentrated clinical focus for CNTY-101 based on unique profile with transformational potential in autoimmune disease; data anticipated in 2025 |
| · | Cash runway estimate extended into fourth quarter of 2026 |
PHILADELPHIA, March 19, 2025 -- Century Therapeutics, Inc. (‘Century’, NASDAQ: IPSC), an innovative biotechnology company developing induced pluripotent stem cell (iPSC)-derived cell therapies in autoimmune disease and cancer, today reported financial results and business highlights for the full year 2024.
“Today we announced a pipeline re-prioritization to streamline resources on advancing candidates that are potentially transformational or best-in-class in diseases with high unmet need. We ended the year with a strong cash position, which we will leverage to achieve meaningful milestones and drive value for all stakeholders as we take the company forward in a new direction,” said Brent Pfeiffenberger, Pharm.D., Chief Executive Officer of Century Therapeutics. “We have made the strategic decision to discontinue the Phase 1 ELiPSE-1 trial early, and we thank the patients, providers and caregivers for their support and participation. We believe CNTY-101 is well-positioned to potentially impact the standard of care meaningfully in B-cell-mediated autoimmune diseases. We are implementing key initiatives to drive toward delivering data in 2025 from the CALiPSO-1 Phase 1 trial, including new site activations and enhanced patient enrollment efforts in both the U.S. and EU, and with further insights from the CARAMEL Phase 1 investigator-initiated clinical trial which is expected to initiate in mid-2025.”
Fourth Quarter 2024 and Recent Highlights
Clinical Pipeline for CNTY-101
| · | Phase 1 CALiPSO-1 trial site expansion in United States and Europe: The first patient in our CALiPSO-1 Phase 1 trial in autoimmune diseases is enrolled and scheduled for dosing in March 2025. Five sites in the U.S. are actively screening patients and Century has increased resourcing for trial site activation and proficient recruitment. The company is also expanding the CALiPSO-1 clinical trial to include additional sites in select European countries and expects enrollment at those sites will initiate in the second half of 2025. |
| · | CARAMEL IIT on track to commence in mid-2025 following CTA approval: In January 2025, the company announced it had entered into an agreement for an investigator-initiated (IIT) Phase 1/2 trial by Professors Georg Schett and Andreas Mackensen of its CD19 CAR-iNK investigational cell therapy candidate CNTY-101 in patients with B-cell mediated autoimmune diseases. The IIT, which is sponsored by the Friedrich-Alexander University Erlangen-Nürnberg, represents the first evaluation by the internationally recognized Schett/Mackensen group of an allogeneic iPSC-derived CD19-directed NK cell therapy for the treatment of autoimmune diseases. The CARAMEL trial is expected to commence in mid-2025 following Clinical Trial Authorization (CTA) approval. |
| · | Early discontinuation of ELiPSE-1 program in late-stage R/R NHL: While the company remains encouraged by the clinical activity and tolerability profile of CNTY-101 in late-stage relapsed-refractory non-Hodgkin’s lymphoma (R/R NHL), the emerging clinical data do not meet the company’s threshold to be considered transformational in this patient population and the program is being discontinued. The company is committed to providing continued treatment access in the ELiPSE-1 trial for patients showing benefit. We believe the ELiPSE-1 data continues to reinforce the potential of CNTY-101 in autoimmune diseases: in addition to encouraging clinical activity in a difficult to treat R/R NHL population and a favorable tolerability profile, translational data also showed evidence of CNTY-101 trafficking to lymph nodes and deep B cell depletion following treatment. The ELiPSE-1 data continues to support proof-of-concept for Allo-Evasion™ and the ability to enable repeat dosing of the company’s cell therapies. Further data is expected to be presented in 2025. |
Preclinical Pipeline
“We look forward to our planned webinar next month where we will dive deeper into the programs we are taking forward. We believe these exciting programs unlock an opportunity to replace current therapies and expand application of cell therapy to areas with serious medical need, starting with what we believe to be our unique ab CD4+/CD8+ CAR-T cells combined with our most advanced Allo-Evasion™ 5.0 technology ,” said Chad Cowan, Ph.D., Chief Scientific Officer of Century Therapeutics. “In the case of CNTY-308 and CNTY-341 in B-cell-mediated diseases, we are aiming for comparable or better performance to approved autologous CAR-T therapies. With our combined expertise in protein engineering, cell differentiation, and manufacturing, we aim to launch allogeneic cell therapies at antibody-like scale and cost. For our solid tumor and non-immune cell programs, this brings the potential to expand access to cell therapies much more broadly.”
| · | Announced pipeline re-prioritization and live webcast on April 22nd: Today the company announced four new prioritized programs anchored by advanced iPSC-derived ’tunable’ CD4+/CD8+ ab T cells with target profiles comparable to autologous CART cells. All four programs are engineered with the company’s proprietary immune evasion technology, Allo-Evasion™ 5.0, designed to enable holistic evasion of T cell, NK cell, and humoral immunity. Management will host a live webcast on Tuesday, April 22nd to discuss each of the prioritized programs in more detail. |
| · | Advancing CNTY-308 toward product candidate selection: CNTY-308 is a CD19-targeted CAR-iT cell therapy engineered with Allo-Evasion™ 5.0 which has demonstrated preclinical characteristics comparable to autologous CD19 CAR-T cells, including proliferation on target engagement, cytokine secretion, cytotoxic elimination of tumor cells, persistence and proliferation on rechallenge. CNTY-308 is being developed for B-cell mediated autoimmune diseases and malignancies. The company expects to initiate IND-enabling studies with CNTY-308 in mid-2025. |
| · | Three additional preclinical programs being taken forward based on their profiles: CNTY-341 is a CD19/CD22 dual-targeted CAR-iT cell therapy engineered with Allo-Evasion™ 5.0 which pairs dual targeting and primary T-cell-like functionality in an allogeneic cell with the goal of providing a differentiated therapy for B cell malignancies. The next program is the company’s first solid tumor CAR iT program exploiting Nectin-4 CAR and other validated targets, engineered with Allo-Evasion™ 5.0 and additional engineering aimed at overcoming the key barriers to success in treating solid tumors. In addition, the company is leveraging its expertise in selective iPSC differentiation to non-immune effector cells with opportunities to potentially accelerate in high-impact therapeutic areas where the company believes its technology and capabilities provide meaningful differentiation. |
Full Year 2024 Financial Results
| · | Cash Position: Cash, cash equivalents, and marketable securities were $220.1 million as of December 31, 2024, as compared to $261.8 million as of December 31, 2023. Net cash used in operations was $110.1 million for the year ended December 31, 2024, compared to net cash used in operations of $88.3 million for the year ended December 31, 2023. The company estimates its cash, cash equivalents, and investments will support operations into the fourth quarter of 2026. |
| · | Collaboration Revenue: Collaboration revenue generated through the company’s collaboration, option, and license agreement with Bristol-Myers Squibb was $6.6 million. | |
| · | Research and Development (R&D) Expenses: R&D expenses were $107.2 million for the year ended December 31, 2024, compared to $92.7 million for the same period in 2023. The increase in R&D expenses is most notably due to increase in research and laboratory costs due to progression of the ELiPSE-1 clinical trial, start-up costs relating to the CALiPSO-1 trial, and manufacturing costs related to the company’s collaboration with FujiFilm Cellular Dynamics, Inc. | |
| · | General and Administrative (G&A) Expenses: G&A expenses were $33.2 million for the year ended December 31, 2024, compared to $34.7 million for the same period in 2023. The decrease was primarily due to a decrease in employee headcount during the 2024 fiscal year. | |
| · | Net Loss: Net loss was $126.6 million for the year ended December 31, 2024, compared to net loss of $136.7 million for the same period in 2023. |
About Century Therapeutics
Century Therapeutics (NASDAQ: IPSC) is harnessing the power of adult stem cells to develop curative cell therapy products for cancer and autoimmune diseases that we believe will allow us to overcome the limitations of first-generation cell therapies. Our genetically engineered, iPSC-derived cell product candidates are designed to specifically target hematologic and solid tumor cancers, with a broadening application to autoimmune diseases. We are leveraging our expertise in cellular reprogramming, genetic engineering, and manufacturing to develop therapies with the potential to overcome many of the challenges inherent to cell therapy and provide a significant advantage over existing cell therapy technologies. We believe our commitment to developing off-the-shelf cell therapies will expand patient access and provide an unparalleled opportunity to advance the course of cancer and autoimmune disease care. For more information on Century Therapeutics, please visit www.centurytx.com.
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of, and made pursuant to the safe harbor provisions of, The Private Securities Litigation Reform Act of 1995. All statements contained in this press release, other than statements of historical facts or statements that relate to present facts or current conditions, including but not limited to, statements regarding our clinical development plans and timelines are forward-looking statements. These statements involve known and unknown risks, uncertainties and other important factors that may cause our actual results, performance, or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements. In some cases, you can identify forward-looking statements by terms such as “may,” “might,” “will,” “should,” “expect,” “plan,” “aim,” “seek,” “anticipate,” “could,” “intend,” “target,” “project,” “contemplate,” “believe,” “estimate,” “predict,” “forecast,” “potential” or “continue” or the negative of these terms or other similar expressions. The forward-looking statements in this press release are only predictions. We have based these forward-looking statements largely on our current expectations and projections about future events and financial trends that we believe may affect our business, financial condition, and results of operations. These forward-looking statements speak only as of the date of this press release and are subject to a number of risks, uncertainties and assumptions, some of which cannot be predicted or quantified and some of which are beyond our control, including, among others: our ability to successfully advance our current and future product candidates through development activities, preclinical studies, and clinical trials; our dependence on the success of our lead product candidate, CNTY-101; our ability to progress CNTY-101 through clinical development; our ability to meet development milestones on anticipated timelines; uncertainties inherent in the results of preliminary data, pre-clinical studies and earlier-stage clinical trials, which may not be predictive of final results or the results of later-stage clinical trials; our ability to obtain FDA clearance of our future IND submissions and commence and complete clinical trials on expected timelines, or at all; our reliance on the maintenance of certain key collaborative relationships for the manufacturing and development of our product candidates; the timing, scope and likelihood of regulatory filings and approvals, including final regulatory approval of our product candidates; the impact of geopolitical issues, banking instability and inflation on our business and operations, supply chain and labor force; the performance of third parties in connection with the development of our product candidates, including third parties conducting our clinical trials as well as third-party suppliers and manufacturers; our ability to successfully commercialize our product candidates and develop sales and marketing capabilities, if our product candidates are approved; our ability to recruit and maintain key members of management and our ability to maintain and successfully enforce adequate intellectual property protection. These and other risks and uncertainties are described more fully in the “Risk Factors” section of our most recent filings with the Securities and Exchange Commission and available at www.sec.gov. You should not rely on these forward-looking statements as predictions of future events. The events and circumstances reflected in our forward-looking statements may not be achieved or occur, and actual results could differ materially from those projected in the forward-looking statements. Moreover, we operate in a dynamic industry and economy. New risk factors and uncertainties may emerge from time to time, and it is not possible for management to predict all risk factors and uncertainties that we may face. Except as required by applicable law, we do not plan to publicly update or revise any forward-looking statements contained herein, whether as a result of any new information, future events, changed circumstances or otherwise.
For More Information:
Century Therapeutics
Morgan Conn, PhD
Chief Financial Officer
investor.relations@centurytx.com
JPA Health
Sarah McCabe
smccabe@jpa.com
Century Therapeutics, Inc
Condensed Balance Sheets
(unaudited, in thousands)
| December 31, | December 31, | |||||||
| Assets | 2024 | 2023 | ||||||
| Current Assets: | ||||||||
| Cash and cash equivalents | $ | 58,441 | $ | 47,324 | ||||
| Short-term investments | 130,851 | 125,414 | ||||||
| Prepaid expenses and other current assets | 4,759 | 4,256 | ||||||
| Total current assets | 194,051 | 176,994 | ||||||
| Property and equipment, net | 62,141 | 71,705 | ||||||
| Operating lease right-of-use assets, net | 28,706 | 20,376 | ||||||
| Long-term investments | 30,818 | 89,096 | ||||||
| Goodwill | - | - | ||||||
| Intangible assets | 34,200 | - | ||||||
| Other long-term assets | 3,300 | 2,520 | ||||||
| Total assets | $ | 353,216 | $ | 360,691 | ||||
| Liabilities, convertible preferred stock, and stockholders' equity | ||||||||
| Current liabilities: | ||||||||
| Accounts payable | $ | 3,075 | $ | 2,741 | ||||
| Accrued expenses and other liabilities | 17,543 | 10,733 | ||||||
| Long-term debt, current | - | - | ||||||
| Deferred revenue, current | 109,164 | 4,372 | ||||||
| Total current liabilities | 129,782 | 17,846 | ||||||
| Operating lease liability, noncurrent | 48,960 | 46,658 | ||||||
| Long-term debt, net | - | - | ||||||
| Other long-term liabilities | - | 56 | ||||||
| Deferred revenue | - | 111,381 | ||||||
| Contingent consideration liability | 8,738 | - | ||||||
| Deferred tax liability | 4,374 | - | ||||||
| Total liabilities | 191,854 | 175,941 | ||||||
| Stockholders' equity | ||||||||
| Common stock | 9 | 6 | ||||||
| Additional paid-in capital | 943,366 | 840,407 | ||||||
| Accumulated deficit | (782,337 | ) | (655,771 | ) | ||||
| Accumulated other comprehensive loss | 324 | 108 | ||||||
| Total stockholders' equity | 161,362 | 184,750 | ||||||
| Total liabilities and stockholders' equity | $ | 353,216 | $ | 360,691 | ||||
| Century Therapeutics, Inc | ||||||||
| Condensed consolidated statements of operations | ||||||||
| (unaudited, in thousands, except share and per share amounts) | ||||||||
| Year Ended | Year Ended | |||||||
| December 31, 2024 | December 31, 2023 | |||||||
| Collaboration Revenue | $ | 6,589 | $ | 2,235 | ||||
| Operating Expenses | ||||||||
| Research and development | 107,244 | 92,710 | ||||||
| General and administrative | 33,155 | 34,706 | ||||||
| In-process research and development | - | 5,000 | ||||||
| Impairment on long-lived assets | - | 16,365 | ||||||
| Impairment of goodwill | 4,327 | - | ||||||
| Total operating expenses | 144,726 | 148,781 | ||||||
| Loss from operations | (138,137 | ) | (146,546 | ) | ||||
| Interest expense | - | (540 | ) | |||||
| Interest income | 13,007 | 12,677 | ||||||
| Other income, net | 354 | (383 | ) | |||||
| Loss before provision for income taxes | (124,776 | ) | (134,792 | ) | ||||
| Provision for income taxes | (1,790 | ) | (1,881 | ) | ||||
| Net Loss | $ | (126,566 | ) | $ | (136,673 | ) | ||
| Unrealized gain (loss) on investments | 153 | 2,602 | ||||||
| Foreign currency translation adjustment gain (loss) | 63 | (32 | ) | |||||
| Comprehensive loss | $ | (126,350 | ) | $ | (134,103 | ) | ||
| Net loss per common share - Basic and Diluted | (1.61 | ) | (2.30 | ) | ||||
| Weighted average common shares outstanding | 78,648,958 | 59,314,389 | ||||||
Exhibit 99.2
Mar 2025 Corporate Overview

2 Forward - looking statements This presentation contains forward - looking statements within the meaning of, and made pursuant to the safe harbor provisions of, The Private Securities Litigation Reform Act of 1995. All statements contained in this presentation, other than statements of historical facts or statements that relate to present fa cts or current conditions, including but not limited to, statements regarding our clinical development plans and timelines and the initial safety and efficacy profiles of CNTY - 101 are f orward - looking statements. These statements involve known and unknown risks, uncertainties and other important factors that may cause our actual results, performance, or achieve men ts to be materially different from any future results, performance or achievements expressed or implied by the forward - looking statements. In some cases, you can identify forward - look ing statements by terms such as “may,” “might,” “will,” “should,” “expect,” “plan,” “aim,” “seek,” “anticipate,” “could,” “intend,” “target,” “project,” “contemplate,” “beli eve ,” “estimate,” “predict,” “forecast,” “potential” or “continue” or the negative of these terms or other similar expressions. The forward - looking statements in this presentation are only predictions. We have based these forward - looking statements largely on our current expectations and projections about future events and financial trends that we believe may affect our business, fi nancial condition, and results of operations. These forward - looking statements speak only as of the date of this presentation and are subject to a number of risks, uncertainties an d assumptions, some of which cannot be predicted or quantified and some of which are beyond our control, including, among others: our ability to successfully advance our current an d future product candidates through development activities, preclinical studies, and clinical trials; our dependence on the success of our lead product candidate, CNTY - 101; our ability to progress CNTY - 101 through clinical development; our ability to meet development milestones on anticipated timelines; uncertainties inherent in the results of pr eli minary data, pre - clinical studies and earlier - stage clinical trials, which may not be predictive of final results or the results of later - stage clinical trials; our ability to obtain FDA cl earance of our future IND submissions and commence and complete clinical trials on expected timelines, or at all; our reliance on the maintenance of certain key collaborative relat ion ships for the manufacturing and development of our product candidates; the timing, scope and likelihood of regulatory filings and approvals, including final regulatory approval of our pro duct candidates; the impact of geopolitical issues, banking instability and inflation on our business and operations, supply chain and labor force; the performance of third parties in c onn ection with the development of our product candidates, including third parties conducting our clinical trials as well as third - party suppliers and manufacturers; our ability to succes sfully commercialize our product candidates and develop sales and marketing capabilities, if our product candidates are approved; our ability to recruit and maintain key members of man agement and our ability to maintain and successfully enforce adequate intellectual property protection. These and other risks and uncertainties are described more fully in the “R isk Factors” section of our most recent filings with the Securities and Exchange Commission and available at www.sec.gov. You should not rely on these forward - looking statements as pred ictions of future events. The events and circumstances reflected in our forward - looking statements may not be achieved or occur, and actual results could differ material ly from those projected in the forward - looking statements. Moreover, we operate in a dynamic industry and economy. New risk factors and uncertainties may emerge from time t o t ime, and it is not possible for management to predict all risk factors and uncertainties that we may face. Except as required by applicable law, we do not plan to publicly up date or revise any forward - looking statements contained herein, whether as a result of any new information, future events, changed circumstances or otherwise.
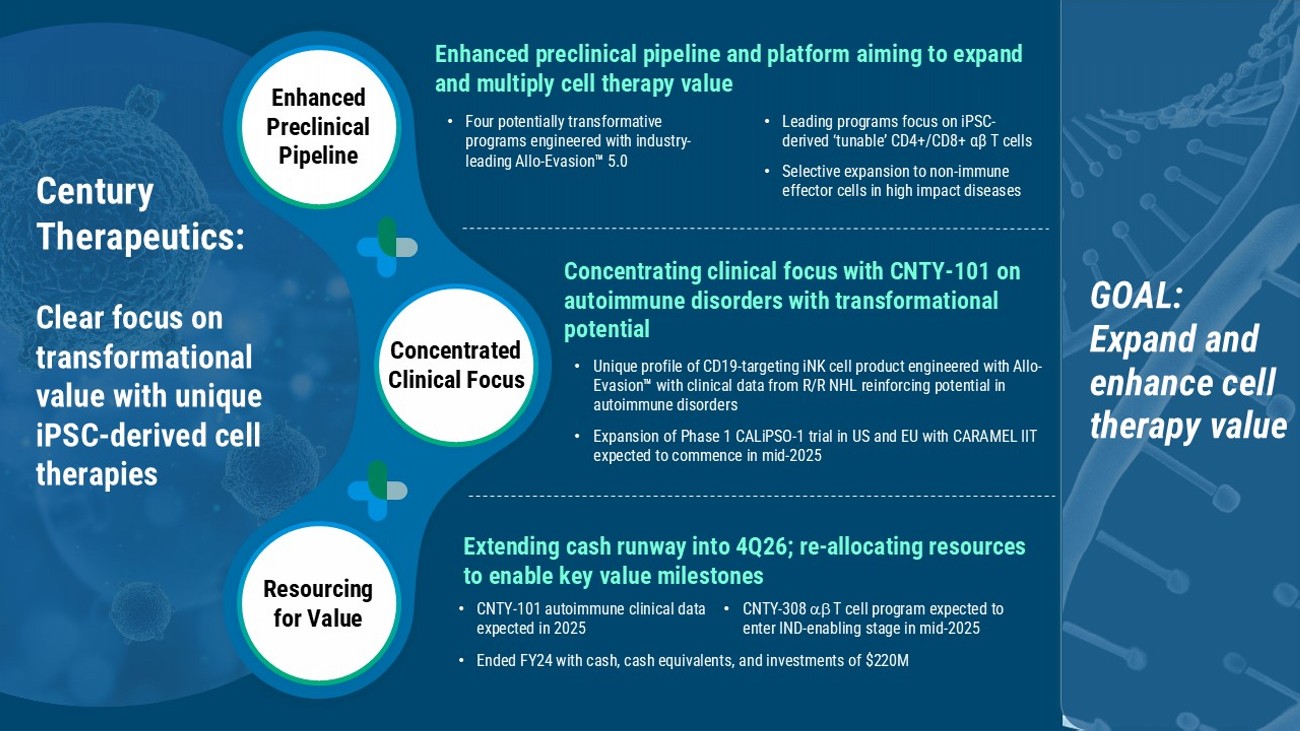
Extending cash runway into 4Q26; re - allocating resources to enable key value milestones Enhanced preclinical pipeline and platform aiming to expand and multiply cell therapy value • Four potentially transformative programs engineered with industry - leading Allo - Evasion 5.0 • Leading programs focus on iPSC - derived ‘tunable’ CD4+/CD8+ αβ T cells • Selective expansion to non - immune effector cells in high impact diseases Enhanced Preclinical Pipeline Concentrated Clinical Focus Resourcing for Value Concentrating clinical focus with CNTY - 101 on autoimmune disorders with transformational potential • Unique profile of CD19 - targeting iNK cell product engineered with Allo - Evasion with clinical data from R/R NHL reinforcing potential in autoimmune disorders • Expansion of Phase 1 CALiPSO - 1 trial in US and EU with CARAMEL IIT expected to commence in mid - 2025 Century Therapeutics: Clear focus on transformational value with unique iPSC - derived cell therapies GOAL: E xpand and enhance cell therapy value • Ended FY24 with cash, cash equivalents, and investments of $220M • CNTY - 308 T cell program expected to enter IND - enabling stage in mid - 2025 • CNTY - 101 autoimmune clinical data expected in 2025
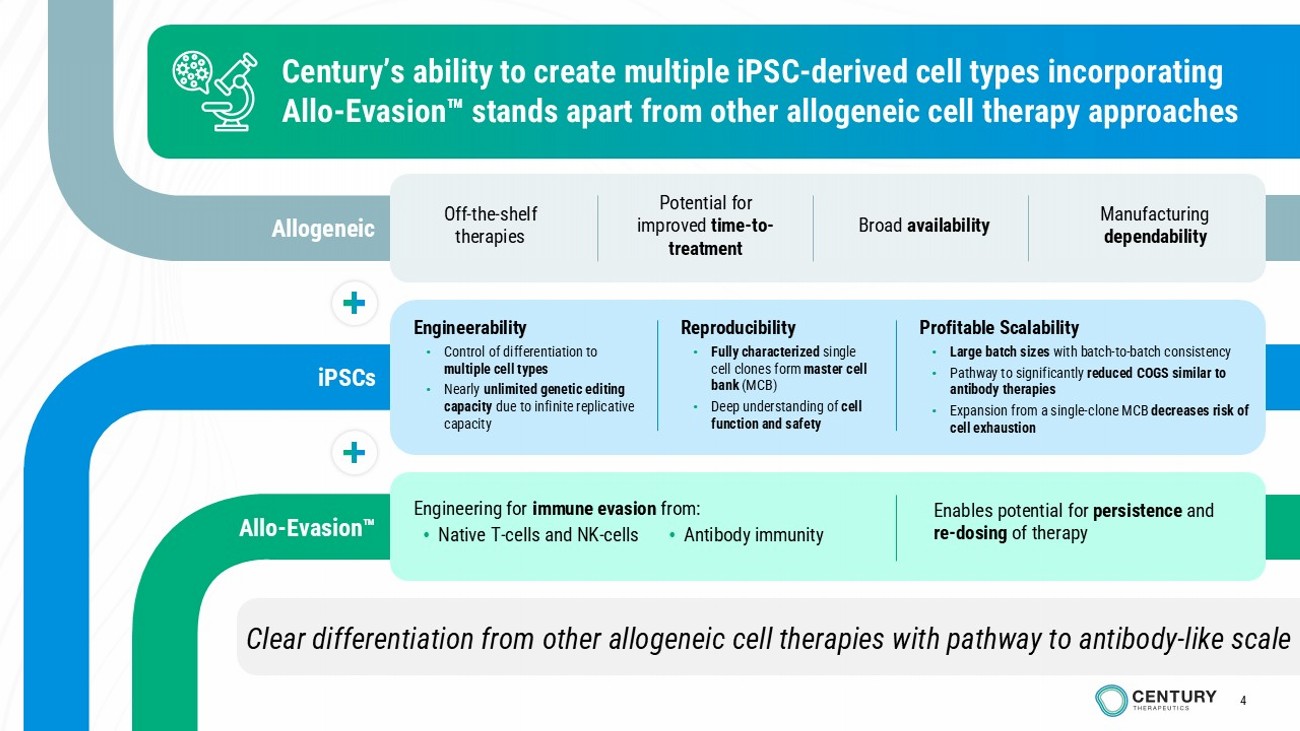
4 Clear differentiation from other allogeneic cell therapies with pathway to antibody - like scale Century’s ability to create multiple iPSC - derived cell types incorporating Allo - Evasion stands apart from other allogeneic cell therapy approaches Engineerability • Control of differentiation to multiple cell types • Nearly unlimited genetic editing capacity due to infinite replicative capacity Reproducibility • Fully characterized single cell clones form master cell bank (MCB) • Deep understanding of cell function and safety Profitable Scalability • Large batch sizes with batch - to - batch consistency • Pathway to significantly reduced COGS similar to antibody therapies • Expansion from a single - clone MCB decreases risk of cell exhaustion Allogeneic Allo - Evasion iPSCs Off - the - shelf therapies Potential for improved time - to - treatment Broad availability Manufacturing dependability Enables potential for persistence and re - dosing of therapy Engineering for immune evasion from: • Native T - cells and NK - cells • Antibody immunity 4
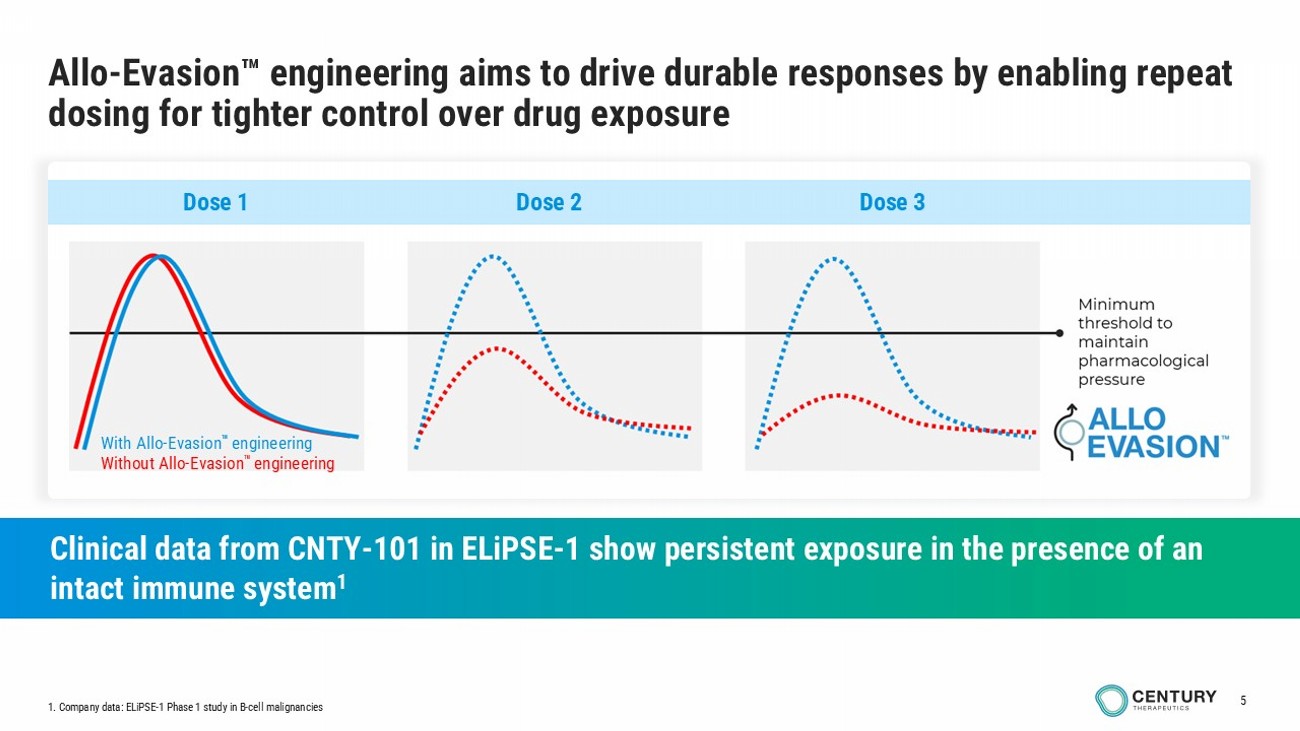
5 Allo - Evasion engineering aims to drive durable responses by enabling repeat dosing for tighter control over drug exposure Clinical data from CNTY - 101 in ELiPSE - 1 show persistent exposure in the presence of an intact immune system 1 With Allo - Evasion engineering Without Allo - Evasion engineering Dose 1 Dose 2 Dose 3 1. Company data: ELiPSE - 1 Phase 1 study in B - cell malignancies
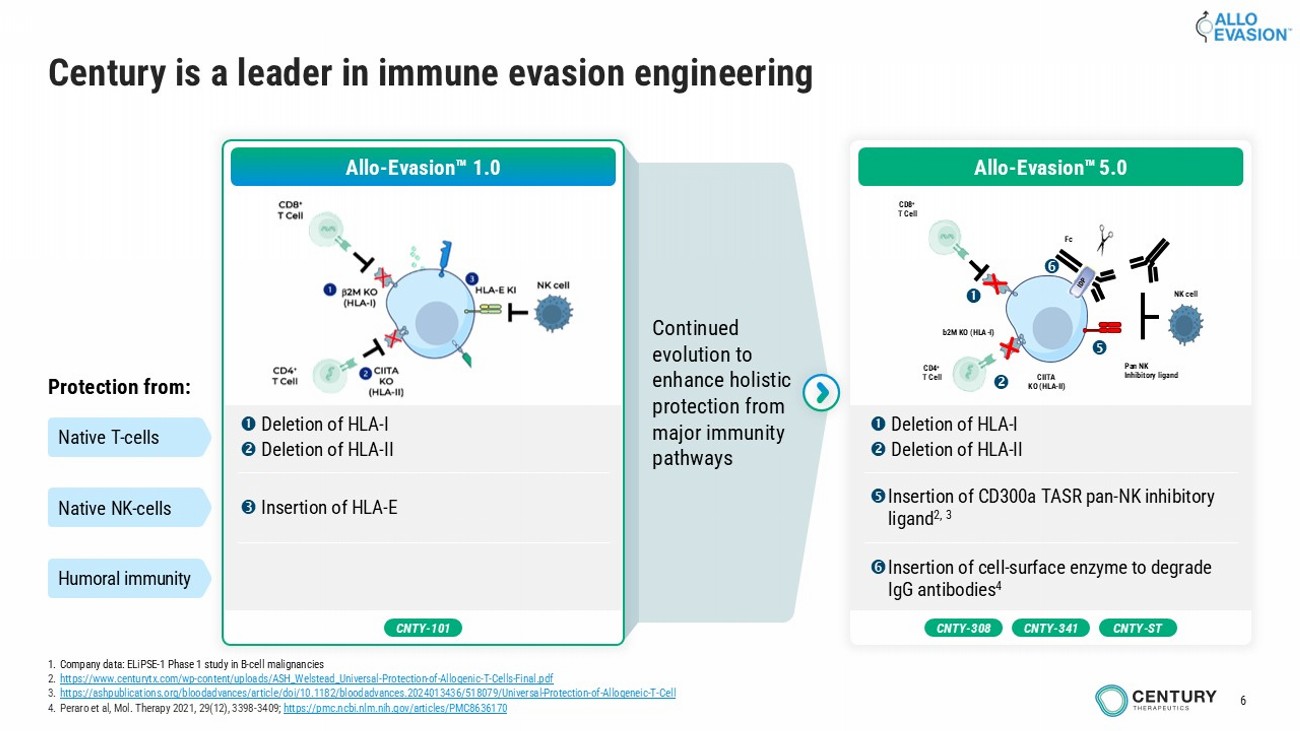
6 Continued evolution to enhance holistic protection from major immunity pathways Century is a leader in immune evasion engineering Allo - Evasion 1.0 Deletion of HLA - I Deletion of HLA - II Insertion of HLA - E Protection from: Native T - cells Native NK - cells Humoral immunity CNTY - 101 1. Company data: ELiPSE - 1 Phase 1 study in B - cell malignancies 2. https://www.centurytx.com/wp - content/uploads/ASH_Welstead_Universal - Protection - of - Allogenic - T - Cells - Final.pdf 3. https://ashpublications.org/bloodadvances/article/doi/10.1182/bloodadvances.2024013436/518079/Universal - Protection - of - Allogeneic - T - Cell 4. Peraro et al, Mol. Therapy 2021, 29(12), 3398 - 3409; https://pmc.ncbi.nlm.nih.gov/articles/PMC8636170 CNTY - 308 CNTY - ST CNTY - 341 Deletion of HLA - I Deletion of HLA - II Insertion of CD300a TASR pan - NK inhibitory ligand 2, 3 Insertion of cell - surface enzyme to degrade IgG antibodies 4 b2M KO (HLA - I) CIITA KO (HLA - II) CD8 + T Cell CD4 + T Cell Pan NK Inhibitory ligand Fc NK cell Allo - Evasion 5.0
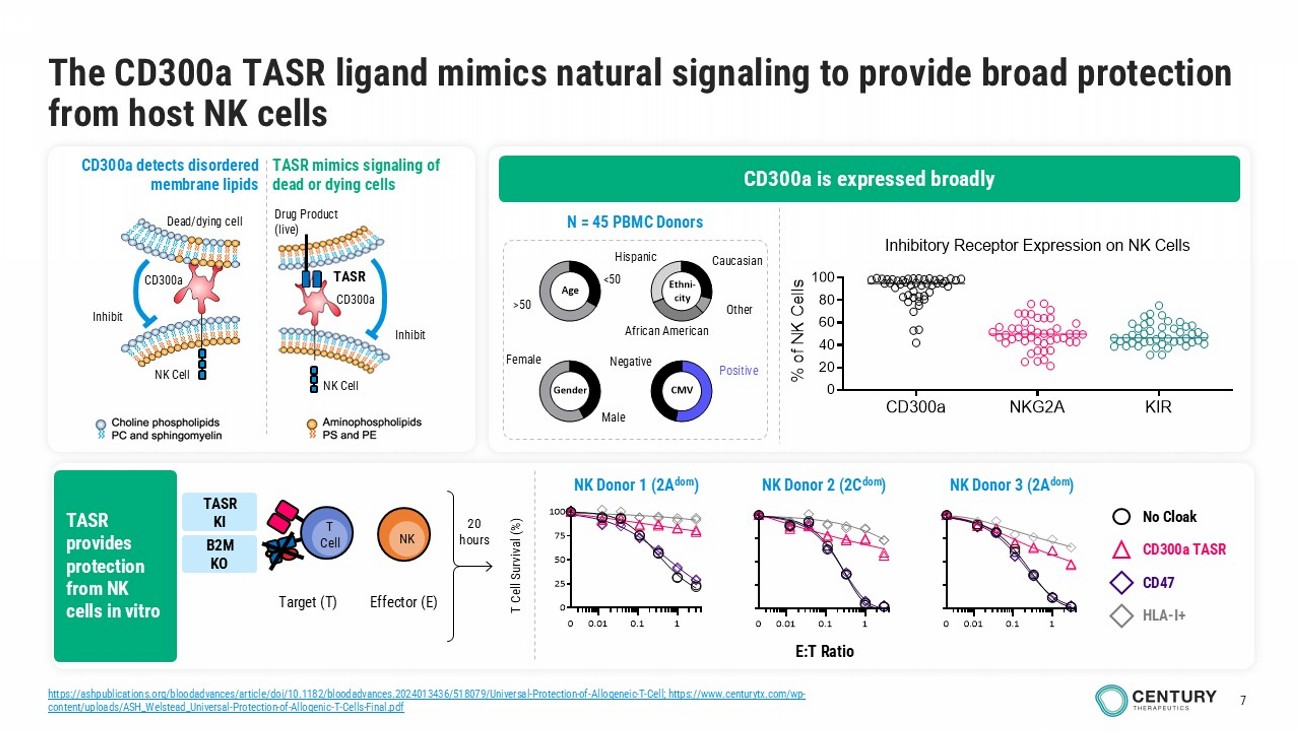
7 The CD300a TASR ligand mimics natural signaling to provide broad protection from host NK cells CD300a detects disordered membrane lipids TASR mimics signaling of dead or dying cells Drug Product (live) Inhibit NK Cell TASR CD300a Dead/dying c ell NK Cell Inhibit CD300a Male Female <50 >50 Gender Age Ethni- city CMV Caucasian Other African American Hispanic Negative Positive N = 45 PBMC Donors 0.01 0.1 10 0.01 0.1 1 0 25 50 75 100 0 E:T Ratio T Cell Survival (%) No Cloak CD300a TASR CD47 HLA - I+ 20 hours Target (T) T Cell NK Effector (E) CD300a NKG2A KIR 0 20 40 60 80 100 Inhibitory Receptor Expression on NK Cells (n = 46 donors) % o f N K C e l l s CD300a is expressed broadly TASR provides protection from NK cells in vitro NK Donor 2 (2C dom ) NK Donor 1 (2A dom ) NK Donor 3 (2A dom ) TASR KI B2M KO https://ashpublications.org/bloodadvances/article/doi/10.1182/bloodadvances.2024013436/518079/Universal - Protection - of - Allogeneic - T - Cell ; https://www.centurytx.com/wp - content/uploads/ASH_Welstead_Universal - Protection - of - Allogenic - T - Cells - Final.pdf
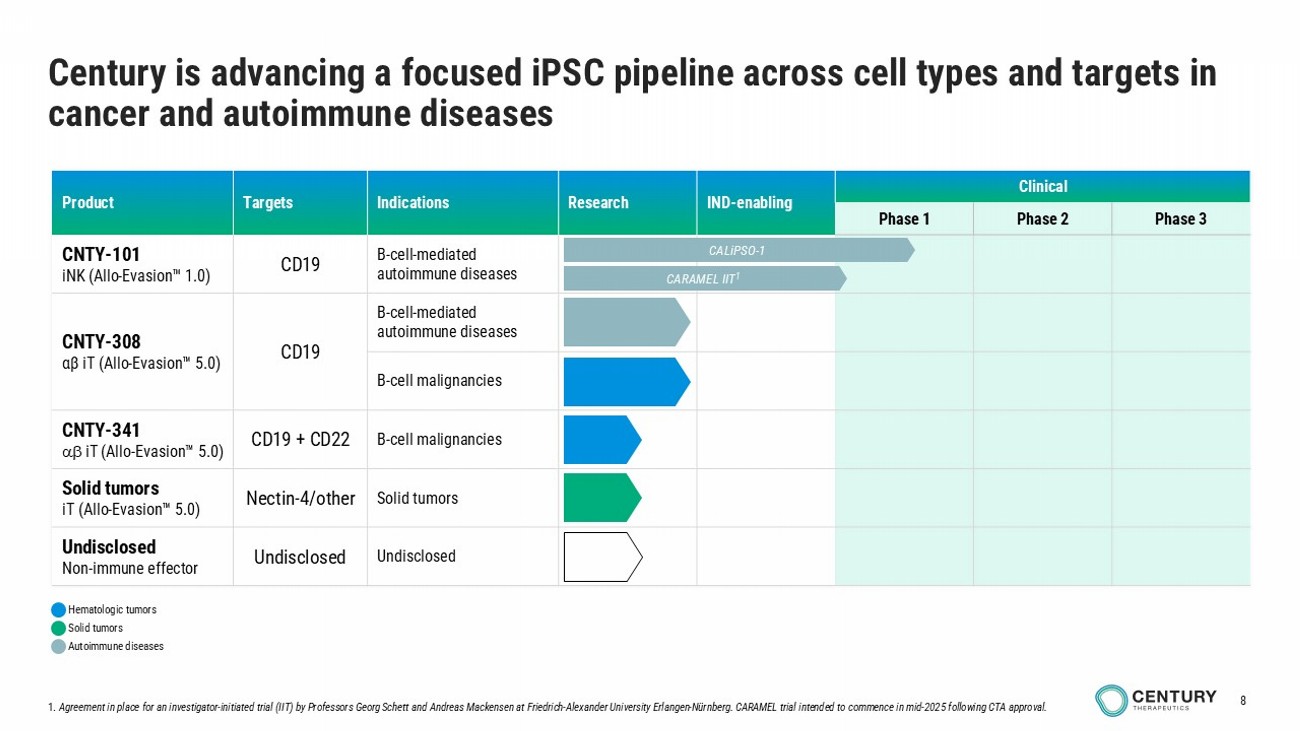
8 Clinical IND - enabling Research Indications Targets Product Phase 3 Phase 2 Phase 1 B - cell - mediated autoimmune diseases CD19 CNTY - 101 iNK ( Allo - Evasion 1.0) B - cell - mediated autoimmune diseases CD19 CNTY - 308 αβ iT ( Allo - Evasion 5.0) B - cell malignancies B - cell malignancies CD19 + CD22 CNTY - 341 iT ( Allo - Evasion 5.0) Solid tumors Nectin - 4/other Solid tumors iT ( Allo - Evasion 5.0) Undisclosed Undisclosed Undisclosed Non - immune effector 1. Agreement in place for an investigator - initiated trial (IIT) by Professors Georg Schett and Andreas Mackensen at Friedrich - Alexander University Erlangen - Nürnberg. CARAMEL trial intended to commence in mid - 2025 following CTA approval. Century is advancing a focused iPSC pipeline across cell types and targets in cancer and autoimmune diseases CALiPSO - 1 CARAMEL IIT 1 Autoimmune diseases Hematologic tumors Solid tumors

CNTY - 101 CAR - iNK cell therapy with Allo - Evasion 1.0
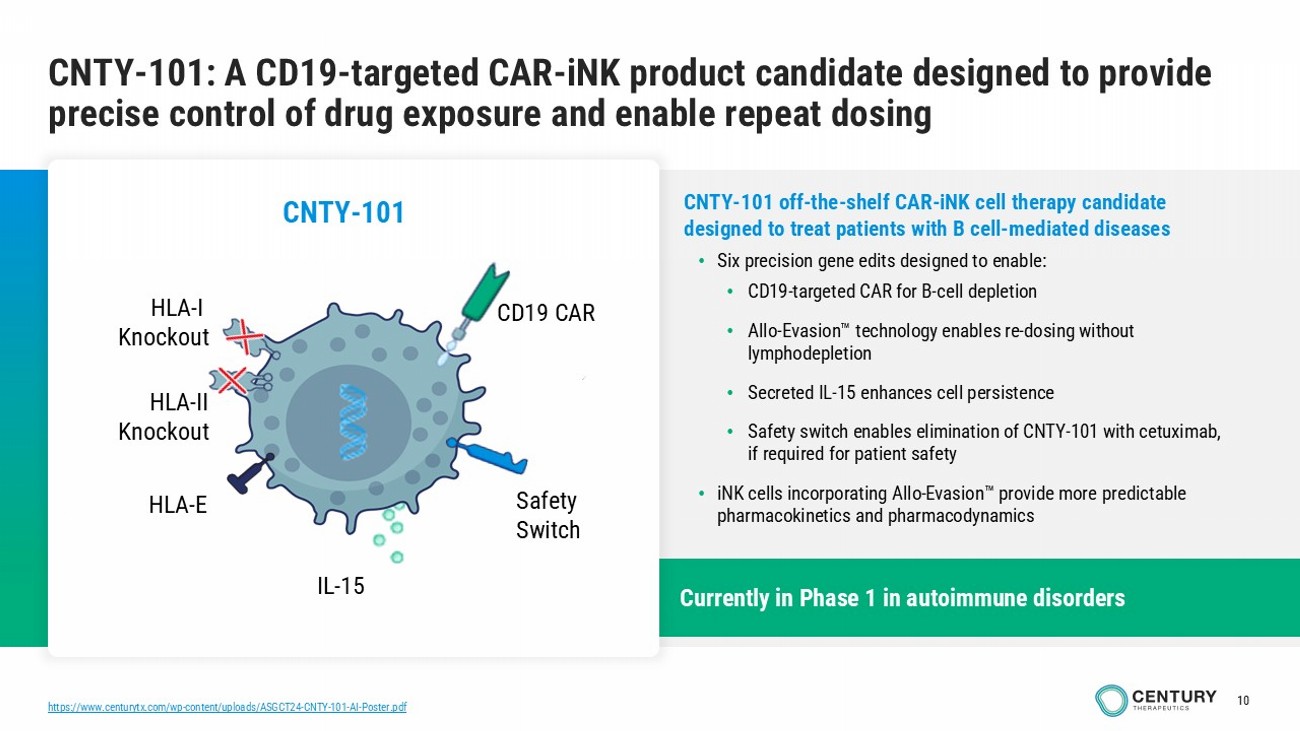
10 Currently in Phase 1 in autoimmune disorders https://www.centurytx.com/wp - content/uploads/ASGCT24 - CNTY - 101 - AI - Poster.pdf CNTY - 101: A CD19 - targeted CAR - iNK product candidate designed to provide precise control of drug exposure and enable repeat dosing CNTY - 101 off - the - shelf CAR - iNK cell therapy candidate designed to treat patients with B cell - mediated diseases • Six precision gene edits designed to enable: • CD19 - targeted CAR for B - cell depletion • Allo - Evasion technology enables re - dosing without lymphodepletion • Secreted IL - 15 enhances cell persistence • Safety switch enables elimination of CNTY - 101 with cetuximab, if required for patient safety • iNK cells incorporating Allo - Evasion provide more predictable pharmacokinetics and pharmacodynamics CNTY - 101 HLA - I Knockout IL - 15 HLA - II Knockout CD19 CAR HLA - E Safety Switch
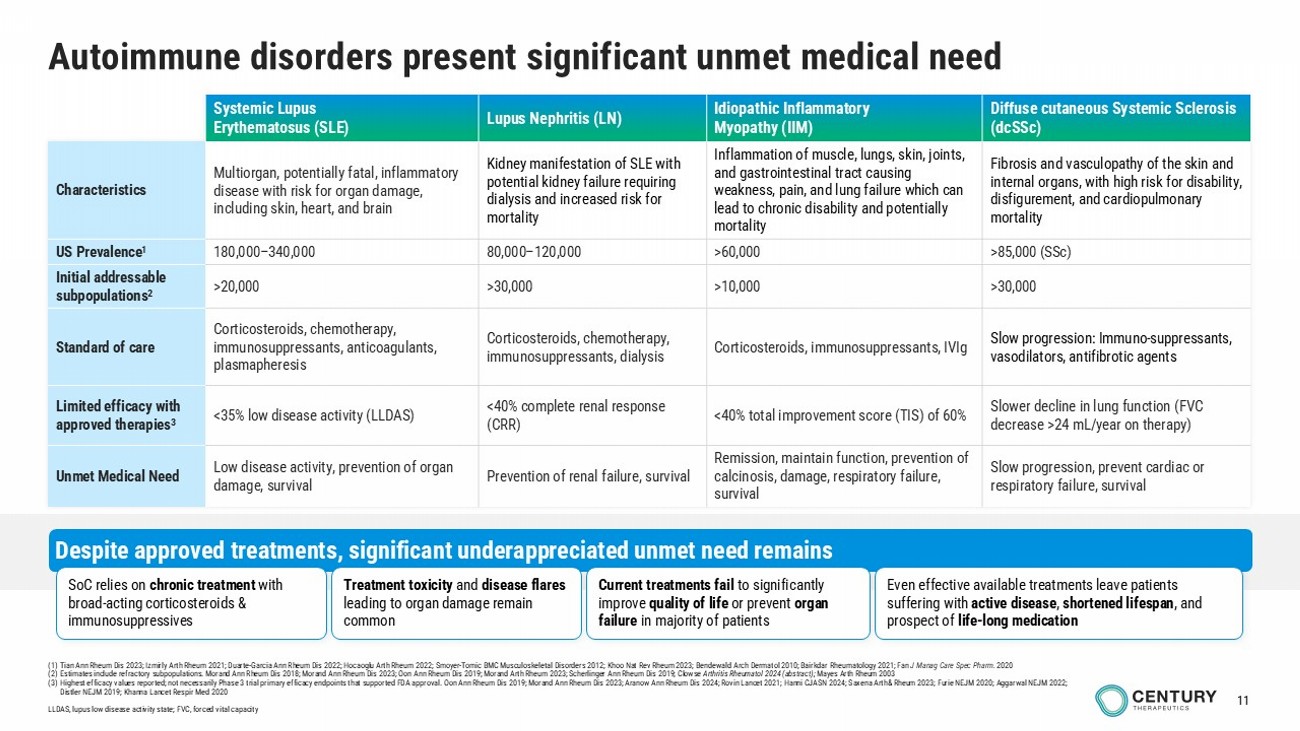
11 Autoimmune disorders present significant unmet medical need Diffuse cutaneous Systemic Sclerosis (dcSSc) Idiopathic Inflammatory Myopathy (IIM) Lupus Nephritis (LN) Systemic Lupus Erythematosus (SLE) Fibrosis and vasculopathy of the skin and internal organs, with high risk for disability, disfigurement, and cardiopulmonary mortality Inflammation of muscle, lungs, skin, joints, and gastrointestinal tract causing weakness, pain, and lung failure which can lead to chronic disability and potentially mortality Kidney manifestation of SLE with potential kidney failure requiring dialysis and increased risk for mortality Multiorgan, potentially fatal, inflammatory disease with risk for organ damage, including skin, heart, and brain Characteristics >85,000 ( SSc ) >60,000 80,000 – 120,000 180,000 – 340,000 US Prevalence 1 >30,000 >10,000 >30,000 >20,000 Initial addressable subpopulations 2 Slow progression: Immuno - suppressants, vasodilators, antifibrotic agents Corticosteroids, immunosuppressants, IVIg Corticosteroids, chemotherapy, immunosuppressants, dialysis Corticosteroids, chemotherapy, immunosuppressants, anticoagulants, plasmapheresis Standard of care Slower decline in lung function (FVC decrease >24 mL/year on therapy) <40% total improvement score (TIS) of 60% <40% complete renal response (CRR) <35% low disease activity (LLDAS) Limited efficacy with approved therapies 3 Slow progression, prevent cardiac or respiratory failure, survival Remission, maintain function, prevention of calcinosis, damage, respiratory failure, survival Prevention of renal failure, survival Low disease activity, prevention of organ damage, survival Unmet Medical Need Despite approved treatments, significant underappreciated unmet need remains Even effective available t reatments leave patients suffering with active disease , shortened lifespan , and prospect of life - long medication SoC relies on chronic treatment with broad - acting corticosteroids & immunosuppressives Treatment toxicity and disease flares leading to organ damage remain common Current treatments fail to significantly improve quality of life or prevent organ failure in majority of patients (1) Tian Ann Rheum Dis 2023; Izmirly Arth Rheum 2021; Duarte - Garcia Ann Rheum Dis 2022; Hocaoglu Arth Rheum 2022 ; Smoyer - Tomic BMC Musculoskeletal Disorders 2012; Khoo Nat Rev Rheum 2023; Bendewald Arch Dermatol 2010; Bairkdar Rheumatology 2021; Fan J Manag Care Spec Pharm. 2020 (2) Estimates include refractory subpopulations. Morand Ann Rheum Dis 2018; Morand Ann Rheum Dis 2023; Oon Ann Rheum Dis 2019; Morand Arth Rheum 2023; Scherlinger Ann Rheum Dis 2019 ; Clowse Arthritis Rheumatol 2024 (abstract); Mayes Arth Rheum 2003 (3) Highest efficacy values reported; not necessarily Phase 3 trial primary efficacy endpoints that supported FDA approval. Oon Ann Rheum Dis 2019; Morand Ann Rheum Dis 2023; Aranow Ann Rheum Dis 2024; Rovin Lancet 2021; Hanni CJASN 2024; Saxena Arth& Rheum 2023; Furie NEJM 2020; Aggarwal NEJM 2022; Distler NEJM 2019; Khanna Lancet Respir Med 2020 LLDAS, lupus low disease activity state; FVC, forced vital capacity

12 Clear opportunity for allogeneic cell therapies to address moderate to severe autoimmune indications by providing long - term, drug - free remission Significant patient population and unmet need • Tens of thousands of patients with unmet need in the US • Heterogeneous nature of patients with autoimmunity supports opportunity for multiple modalities within and across indications • Treatments needed to resolve inflammation , prevent organ failure, normalize lifespan , and avoid toxicity of life - long medication Opportunity to deliver transformational efficacy • Dramatically improve upon standard of care • SLE: LLDAS achievement – predictor for reduction of damage accrual • LN: C omplete renal response (CRR) • SSc : High % CRISS, FVC stabilization • IIM: High % TIS • Optimal outcome: drug - free remission Compelling evidence for benefit from deep depletion of pathogenic B - cells • Autologous anti - CD19 CAR - T cell therapies demonstrate potential for long - term drug - free remission • Unmet c hallenges include safety (CRS, ICANS, neutropenia, B cell aplasia), logistics, and product availability • Emerging data for allogeneic cell therapies 2 demonstrate potential for transformative impact and may address above challenges 1. Mackensen Nature Medicine 2022 doi.org/10.1038/s41591 - 022 - 02017 - 5 , Muller NEJM 2024 doi /full/10.1056/NEJMoa2308917 , Muller ASH 2024 doi.org/10.1182/blood - 2024 - 194525, Sheikh Arthritis Rheumatol . 2024 2. Yu Arthritis Rheumatol . 2024; Goulding Arthritis Rheumatol . 2024, Wang Cell 2024 doi.org/10.1016/j.cell.2024.06.027 CRISS: Composite Response Index for Clinical Trials in Early Diffuse Systemic Sclerosis

13 Allogeneic iPSC • Available ‘off - the - shelf ’ • No patient apheresis required • No manufacturing wait time • Batch - to - batch consistency • Platform enables lower COGs than donor - derived or autologous NK cells • Killing potency (≥ primary CAR - T) leads to deep B - cell depletion 1 • Trafficking to secondary lymphoid tissues and marrow favors pathogenic B - cell targeting • Short - lived, more predictable pharmacokinetics and pharmacodynamics • Manageable safety profile, w ell - tolerated in ELiPSE - 1 Allo - Evasion • Avoiding host immune rejection • Ability to repeat dose without continued lymphodepletion • Ability to re - treat, if needed CNTY - 101 is a differentiated autoimmune disease treatment: Allogeneic iPSC CAR iNK cell therapy with Allo - Evasion 1. https://www.centurytx.com/wp - content/uploads/ASH_Chin_Natural - Killer - GD - Cells - Final.pdf Tighter control over drug exposure: B - cell depletion without prolonged B - cell aplasia
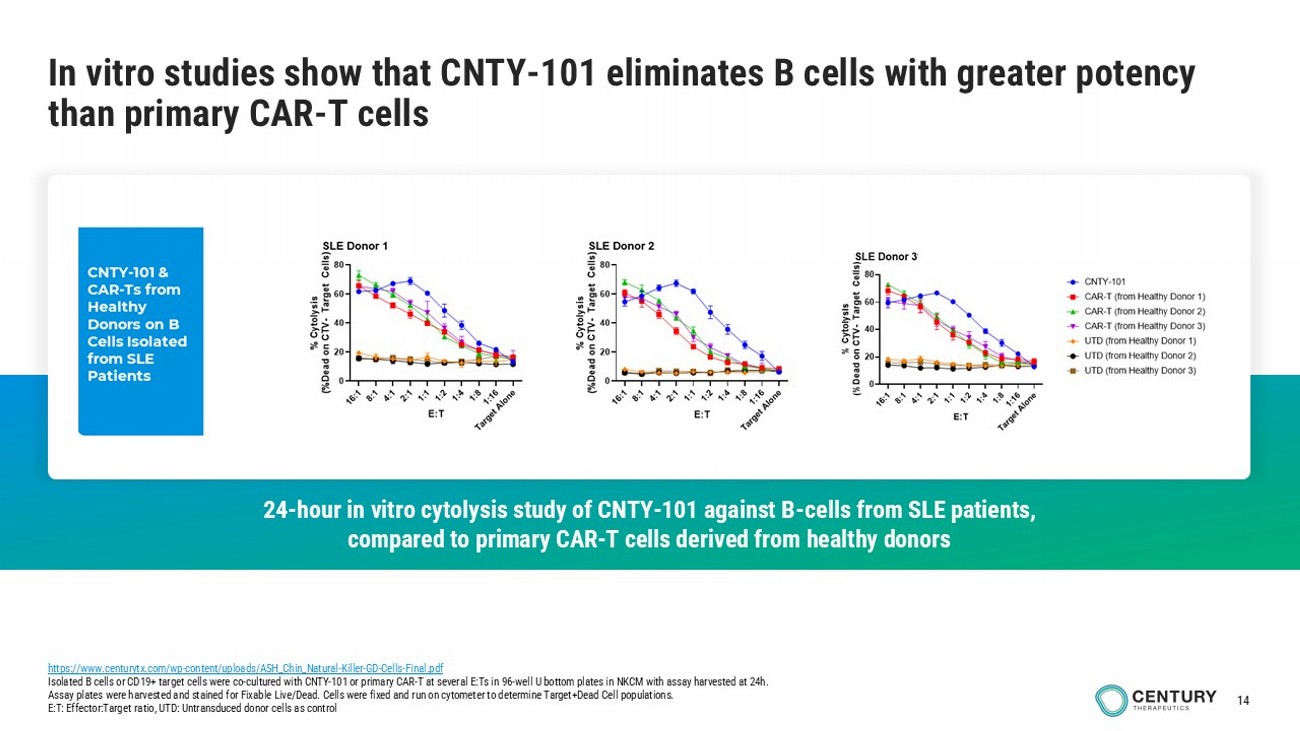
14 In vitro studies show that CNTY - 101 eliminates B cells with greater potency than primary CAR - T cells https://www.centurytx.com/wp - content/uploads/ASH_Chin_Natural - Killer - GD - Cells - Final.pdf Isolated B cells or CD19+ target cells were co - cultured with CNTY - 101 or primary CAR - T at several E:Ts in 96 - well U bottom plate s in NKCM with assay harvested at 24h. Assay plates were harvested and stained for Fixable Live/Dead. Cells were fixed and run on cytometer to determine Target+Dead Cell populations. E:T: Effector:Target ratio, UTD: Untransduced donor cells as control 24 - hour in vitro cytolysis study of CNTY - 101 against B - cells from SLE patients, compared to primary CAR - T cells derived from healthy donors
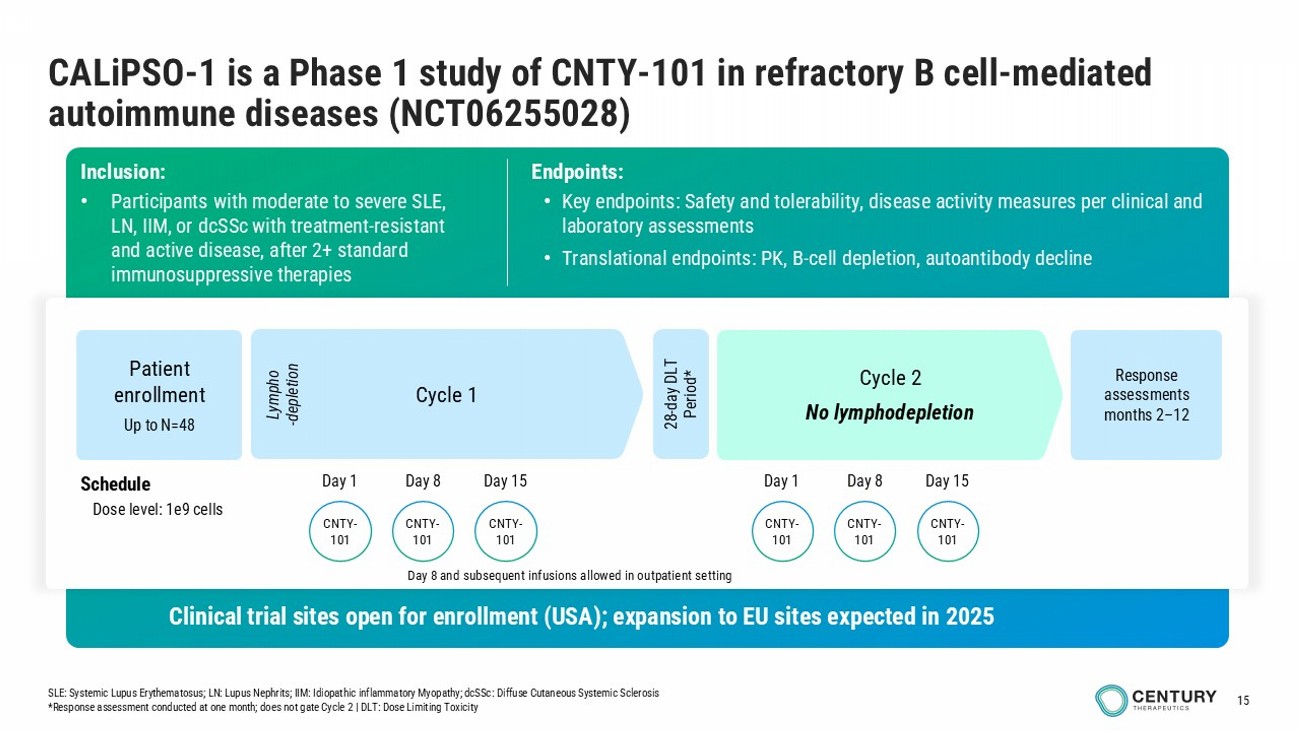
15 CALiPSO - 1 is a Phase 1 study of CNTY - 101 in refractory B cell - mediated autoimmune diseases (NCT06255028) infusions in outpatient setting Inclusion: • Participants with moderate to severe SLE , LN, IIM, or dcSSc with treatment - resistant and active disease, after 2+ standard immunosuppressive therapies Endpoints: • Key endpoints: Safety and tolerability, disease activity measures per clinical and laboratory assessments • Translational endpoints: PK, B - cell depletion, autoantibody decline Cycle 1 Cycle 2 No lymphodepletion Schedule Dose level: 1e9 cells Lympho - depletion 28 - day DLT Period* Patient enrollment Up to N= 48 Response assessments months 2 – 12 Clinical trial sites open for enrollment (USA); expansion to EU sites expected in 2025 SLE: Systemic Lupus Erythematosus; LN: Lupus Nephrits ; IIM: Idiopathic inflammatory Myopathy; dcSSc : Diffuse Cutaneous Systemic Sclerosis *Response assessment conducted at one month; does not gate Cycle 2 | DLT: Dose Limiting Toxicity CNTY - 101 CNTY - 101 CNTY - 101 Day 1 Day 8 Day 15 CNTY - 101 CNTY - 101 CNTY - 101 Day 1 Day 8 Day 15 Day 8 and subsequent infusions allowed in outpatient setting

16 CNTY - 101 ELiPSE - 1 first - in - human study: Initial clinical experience in relapsed/refractory B - cell lymphoma validates Century’s IPSC platform Heavily pre - treated patient population (n=23 safety; n=22 efficacy) • Median 4 prior lines (range 2 - 6); 48% (11/23) of patients received prior CART Favorable initial safety and tolerability profile (n=23) • No dose - limiting toxicities (DLTs); no events of graft - versus - host disease (GvHD) • Majority of participants received CNTY - 101 infusions in an outpatient setting • In DL 3B and 4B (n=9), No ICANs; 3 patients (33%) had G1 or G2 cytokine release syndrome (CRS) Activity Profile in Relapsed / Refractory Aggressive BCL • ORR for DL 3B and 4B (n=9) was 77% (7/9) and complete response rate was 22% (2/9) Source: Company data available as of March 1, 2025 Emerging data reinforce potential of CNTY - 101 in autoimmune diseases at targeted dose levels (DL3B, 4B) • CNTY - 101 cells were detected in lymph node tumor biopsies early post - treatment • CNTY - 101 treatment demonstrates deep B cell depletion • CNTY - 101 infusions showed similar exposure in the presence or absence of endogenous lymphocytes
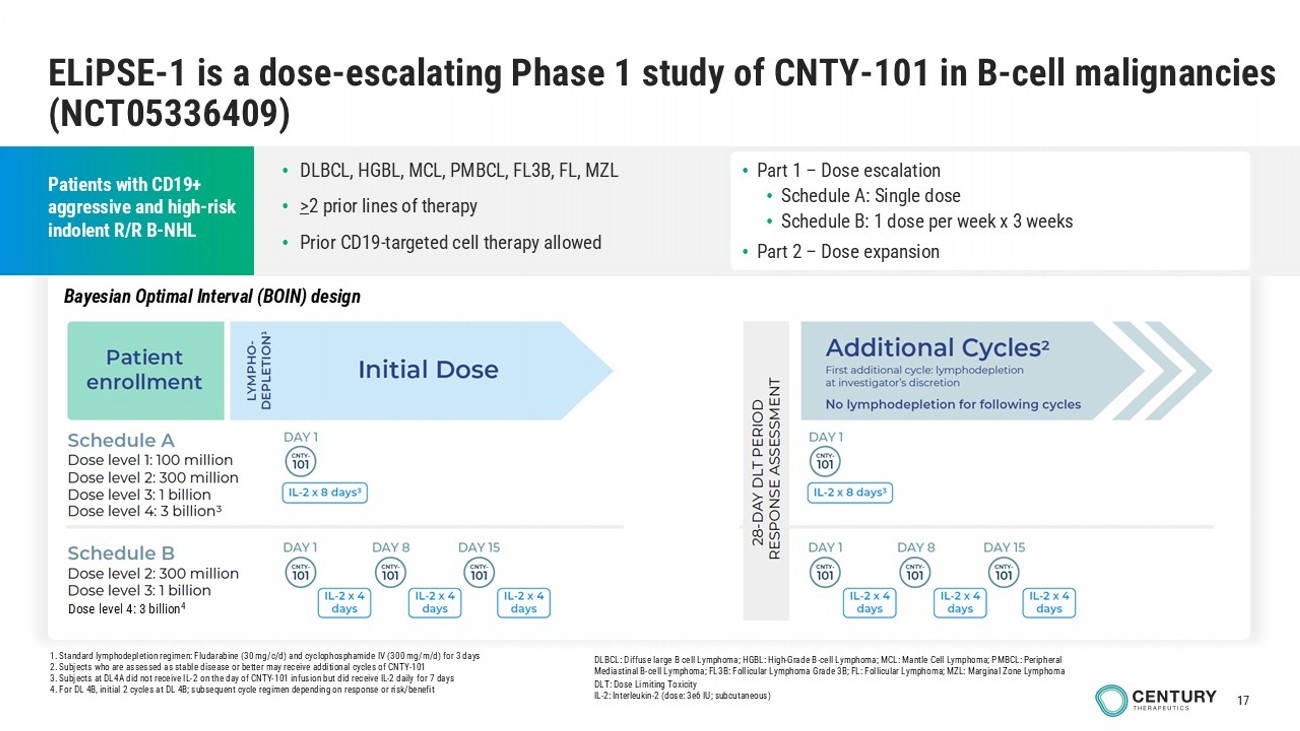
17 1. Standard lymphodepletion regimen: Fludarabine (30 mg/c/d) and cyclophosphamide IV (300 mg/m/d) for 3 days 2. Subjects who are assessed as stable disease or better may receive additional cycles of CNTY - 101 3. Subjects at DL4A did not receive IL - 2 on the day of CNTY - 101 infusion but did receive IL - 2 daily for 7 days 4. For DL 4B, initial 2 cycles at DL 4B; subsequent cycle regimen depending on response or risk/benefit ELiPSE - 1 is a dose - escalating Phase 1 study of CNTY - 101 in B - cell malignancies (NCT05336409) Patients with CD19+ aggressive and high - risk indolent R/R B - NHL • Part 1 – Dose escalation • Schedule A: Single dose • Schedule B: 1 dose per week x 3 weeks • Part 2 – Dose expansion DLBCL: Diffuse large B cell Lymphoma; HGBL: High - Grade B - cell Lymphoma; MCL: Mantle Cell Lymphoma; PMBCL: Peripheral Mediastinal B - cell Lymphoma; FL3B: Follicular Lymphoma Grade 3B; FL: Follicular Lymphoma; MZL: Marginal Zone Lymphoma DLT: Dose Limiting Toxicity IL - 2: Interleukin - 2 (dose: 3e6 IU; subcutaneous) • DLBCL, HGBL, MCL, PMBCL, FL3B, FL, MZL • > 2 prior lines of therapy • Prior CD19 - targeted cell therapy allowed Dose level 4: 3 billion 4 Bayesian Optimal Interval (BOIN) design
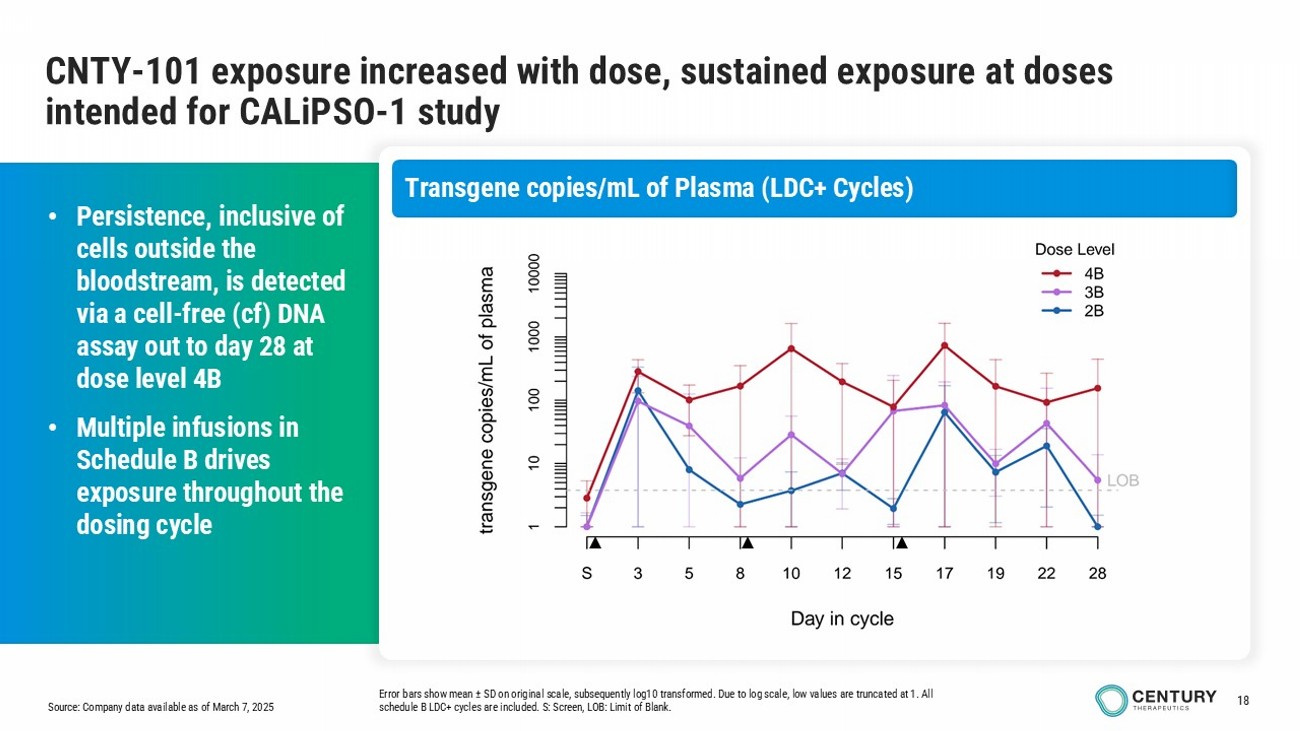
18 Source: Company data available as of March 7, 2025 CNTY - 101 exposure increased with dose, sustained exposure at doses intended for CALiPSO - 1 study Error bars show mean ± SD on original scale, subsequently log10 transformed. Due to log scale, low values are truncated at 1. All schedule B LDC+ cycles are included. S: Screen, LOB: Limit of Blank. • Persistence, inclusive of cells outside the bloodstream, is detected via a cell - free ( cf ) DNA assay out to day 28 at dose level 4B • Multiple infusions in Schedule B drives exposure throughout the dosing cycle Transgene copies/mL of Plasma (LDC+ Cycles)
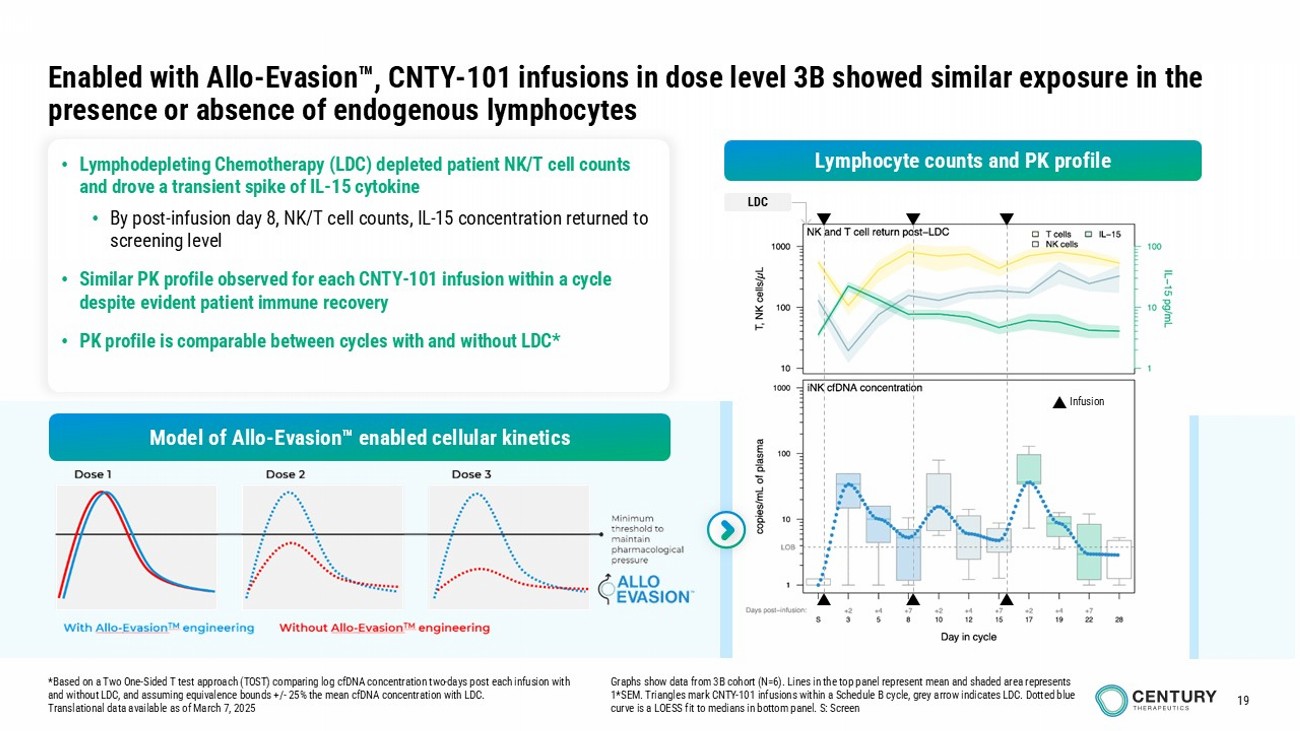
19 Infusion LDC • Lymphodepleting Chemotherapy (LDC) depleted patient NK/T cell counts and drove a transient spike of IL - 15 cytokine • By post - infusion day 8, NK/T cell counts, IL - 15 concentration returned to screening level • Similar PK profile observed for each CNTY - 101 infusion within a cycle despite evident patient immune recovery • PK profile is comparable between cycles with and without LDC* Enabled with Allo - Evasion , CNTY - 101 infusions in dose level 3B showed similar exposure in the presence or absence of endogenous lymphocytes * Based on a Two One - Sided T test approach (TOST) comparing log cfDNA concentration two - days post each infusion with and without LDC, and assuming equivalence bounds +/ - 25% the mean cfDNA concentration with LDC. Translational data available as of March 7, 2025 Graphs show data from 3B cohort (N=6). Lines in the top panel represent mean and shaded area represents 1*SEM. Triangles mark CNTY - 101 infusions within a Schedule B cycle, grey arrow indicates LDC. Dotted blue curve is a LOESS fit to medians in bottom panel. S: Screen Lymphocyte counts and PK profile Model of Allo - Evasion enabled cellular kinetics
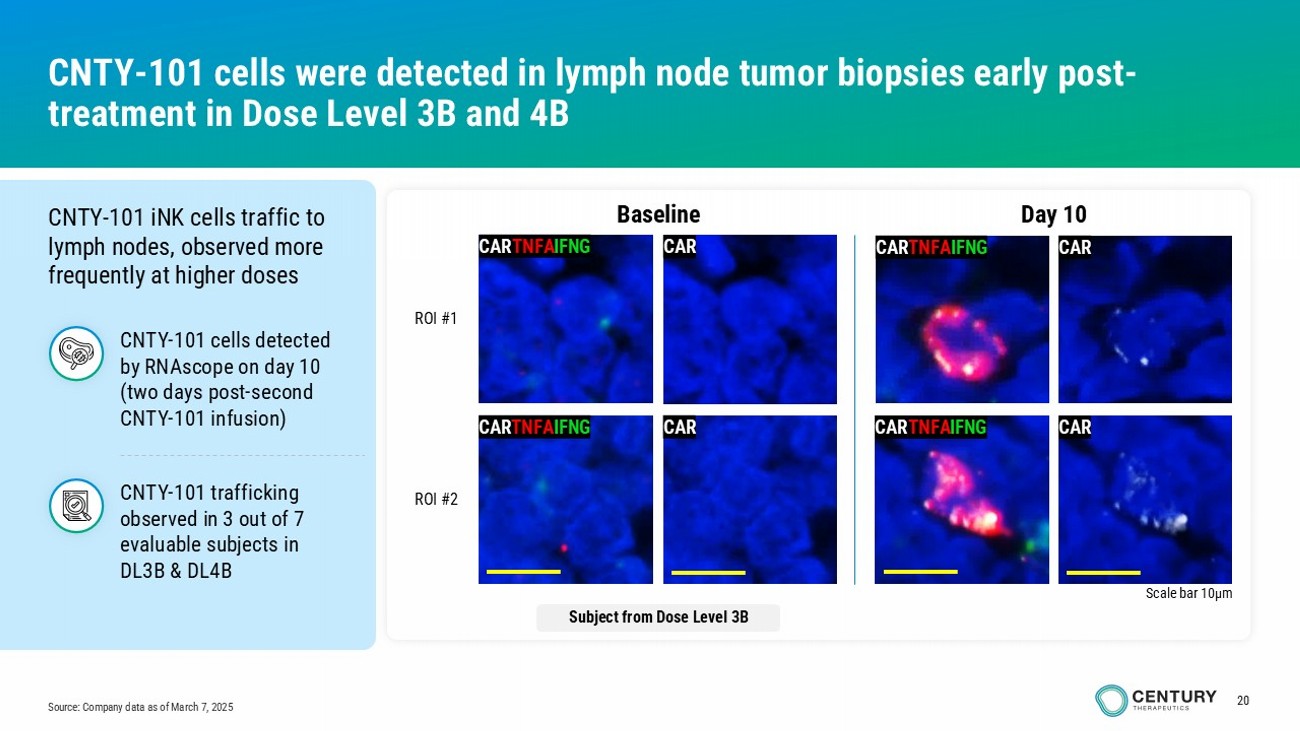
20 Source: Company data as of March 7, 2025 CNTY - 101 cells were detected in lymph node tumor biopsies early post - treatment in Dose Level 3B and 4B CNTY - 101 iNK cells traffic to lymph nodes, observed more frequently at higher doses CAR TNFA IFNG CAR TNFA IFNG CAR CAR Day 10 Baseline CAR TNFA IFNG Scale bar 10µm CAR CAR TNFA IFNG CAR ROI #1 ROI #2 Subject from Dose Level 3B CNTY - 101 trafficking o bserved i n 3 out of 7 evaluable subjects in DL3B & DL4B CNTY - 101 cells detected by RNAscope on day 10 (two days post - second CNTY - 101 infusion)
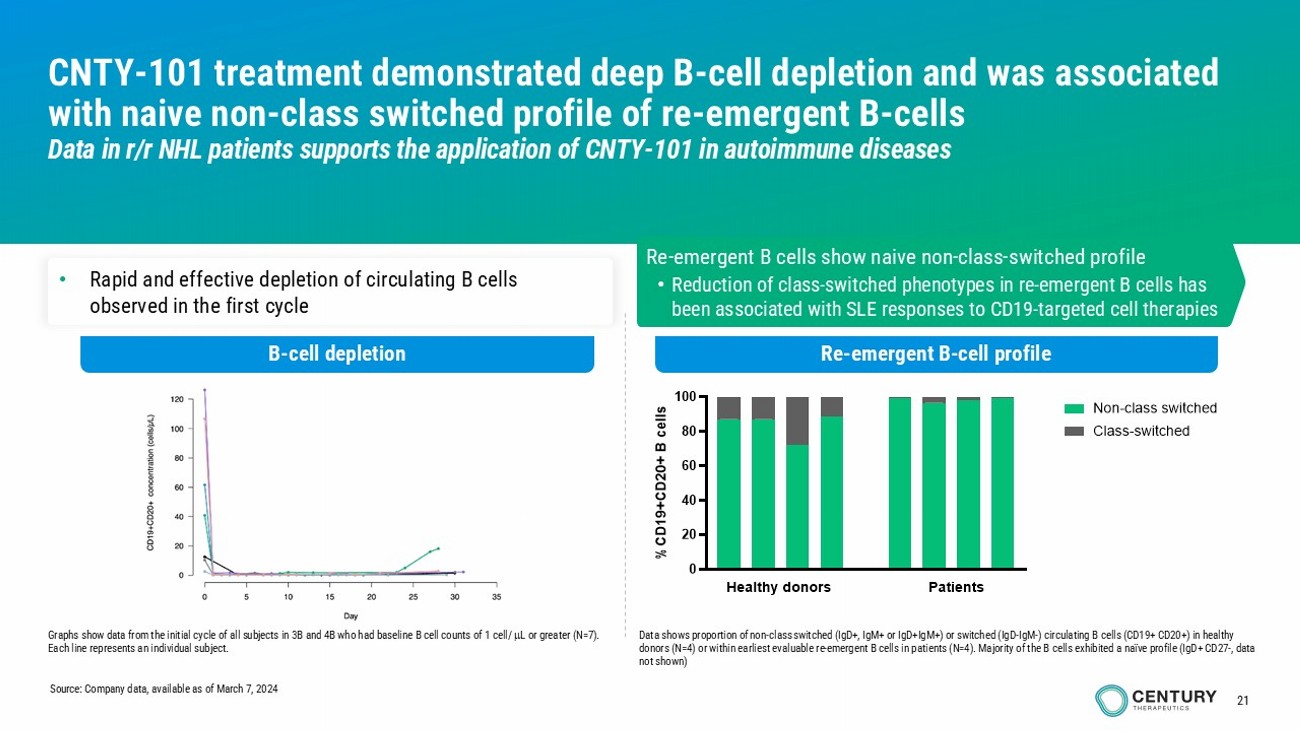
21 CNTY - 101 treatment demonstrated deep B - cell depletion and was associated with naive non - class switched profile of re - emergent B - cells Data in r/r NHL patients supports the application of CNTY - 101 in autoimmune diseases B - cell depletion Re - emergent B - cell profile • Rapid and effective depletion of circulating B cells observed in the first cycle Re - emergent B cells show naive non - class - switched profile • Reduction of class - switched phenotypes in re - emergent B cells has been associated with SLE responses to CD19 - targeted cell therapies Graphs show data from the initial cycle of all subjects in 3B and 4B who had baseline B cell counts of 1 cell/ L or greater (N=7). Each line represents an individual subject. Data shows proportion of non - class switched (IgD+, IgM+ or IgD+IgM+) or switched (IgD - IgM - ) circulating B cells (CD19+ CD20+) in healthy donors (N=4) or within earliest evaluable re - emergent B cells in patients (N=4). Majority of the B cells exhibited a naïve profi le (IgD+ CD27 - , data not shown) Source: Company data, available as of March 7, 2024

CNTY - 308 CAR αβ - iT cell with Allo - Evasion 5.0
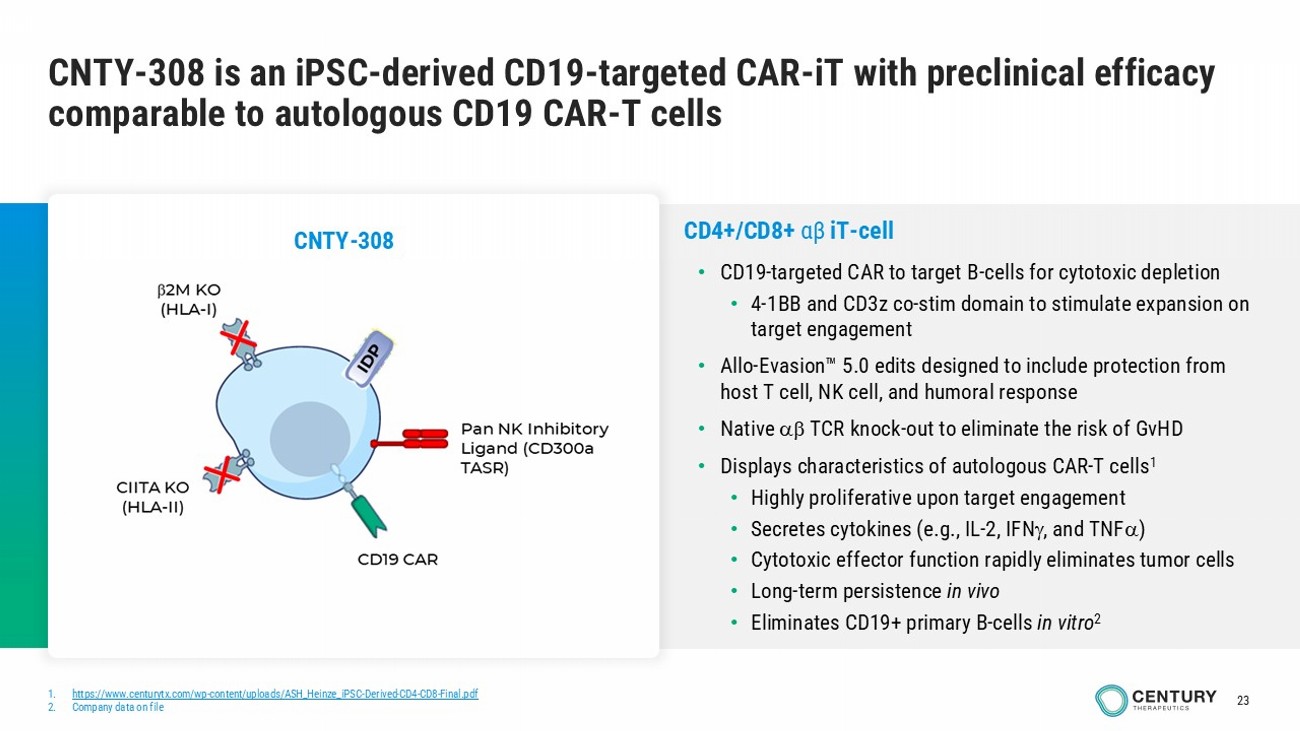
23 CNTY - 308 is an iPSC - derived CD19 - targeted CAR - iT with preclinical efficacy comparable to autologous CD19 CAR - T cells 1. https://www.centurytx.com/wp - content/uploads/ASH_Heinze_iPSC - Derived - CD4 - CD8 - Final.pdf 2. Company data on file CD4+/CD8+ αβ iT - cell • CD19 - targeted CAR to target B - cells for cytotoxic depletion • 4 - 1BB and CD3z co - stim domain to stimulate expansion on target engagement • Allo - Evasion 5.0 edits designed to include protection from host T cell, NK cell, and humoral response • Native TCR knock - out to eliminate the risk of GvHD • Displays characteristics of autologous CAR - T cells 1 • Highly proliferative upon target engagement • Secretes cytokines (e.g., IL - 2, IFN , and TNF ) • Cytotoxic effector function rapidly eliminates tumor cells • Long - term persistence in vivo • Eliminates CD19+ primary B - cells in vitro 2 CNTY - 308
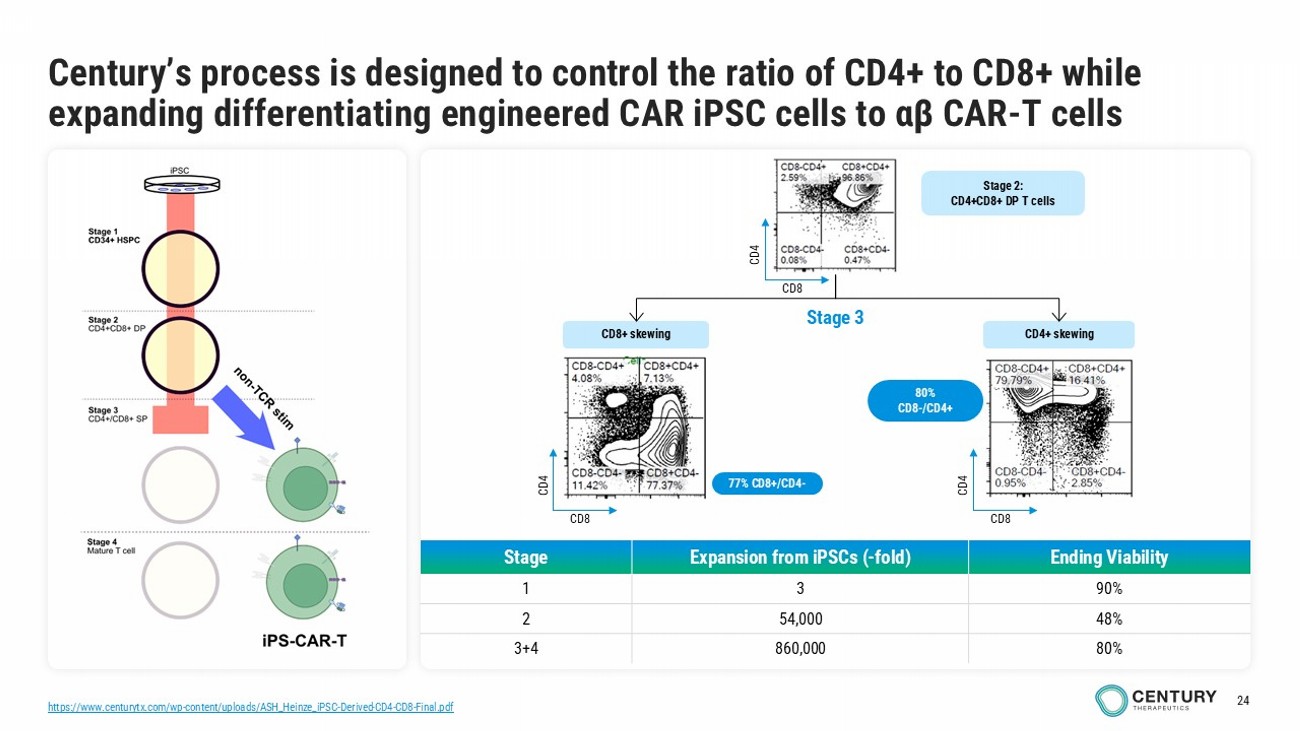
24 https://www.centurytx.com/wp - content/uploads/ASH_Heinze_iPSC - Derived - CD4 - CD8 - Final.pdf Century’s process is designed to control the ratio of CD4+ to CD8+ while expanding differentiating engineered CAR iPSC cells to αβ CAR - T cells Ending Viability Expansion from iPSCs ( - fold) Stage 90% 3 1 48% 54,000 2 80% 860,000 3+4 77% CD8+/CD4 - CD8+ skewing CD4+ skewing Stage 3 Stage 2: CD4+CD8+ DP T cells CD8 CD4 CD8 CD4 CD8 CD4 80% CD8 - /CD4+
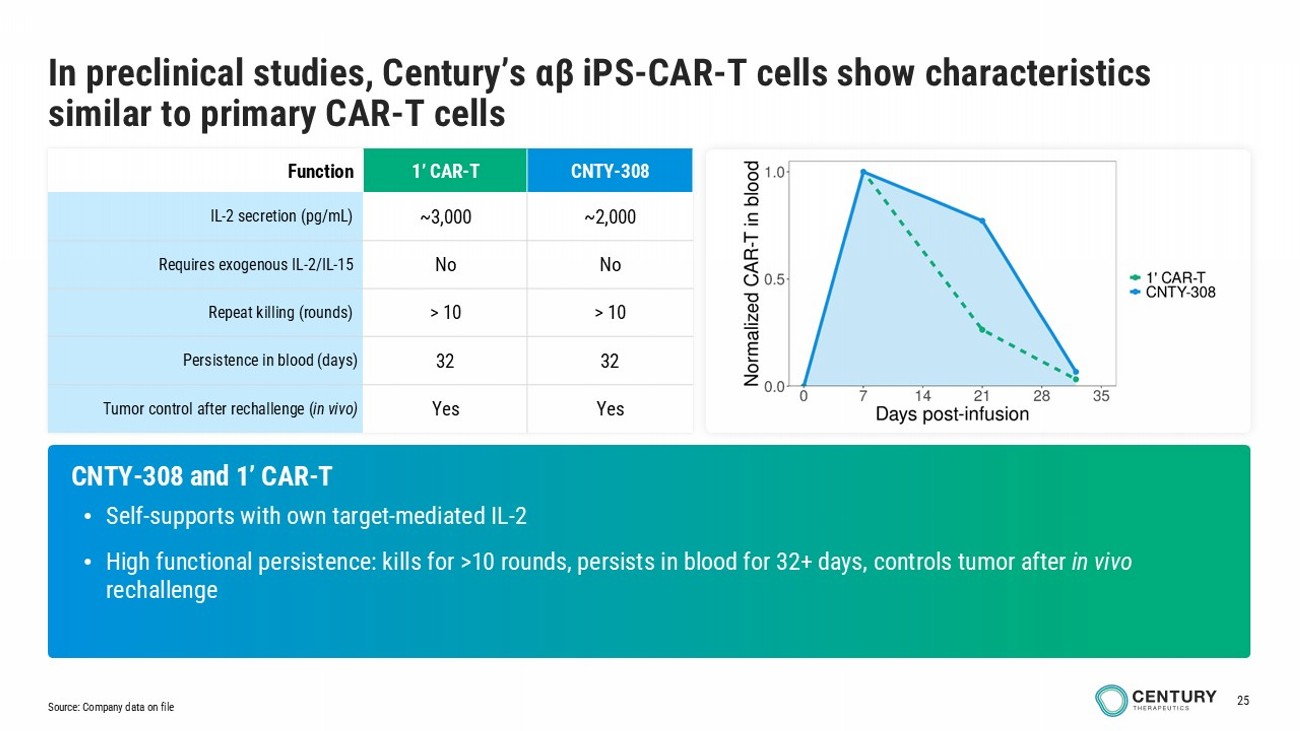
25 CNTY - 308 1’ CAR - T Function ~2,000 ~3,000 IL - 2 secretion (pg/mL) No No Requires exogenous IL - 2/IL - 15 > 10 > 10 Repeat killing (rounds) 32 32 Persistence in blood (days) Yes Yes Tumor control after rechallenge ( in vivo) CNTY - 308 and 1’ CAR - T • Self - supports with own target - mediated IL - 2 • High functional persistence: kills for >10 rounds, persists in blood for 32+ days, controls tumor after in vivo rechallenge Source: Company data on file In preclinical studies, Century’s αβ iPS - CAR - T cells show characteristics similar to primary CAR - T cells
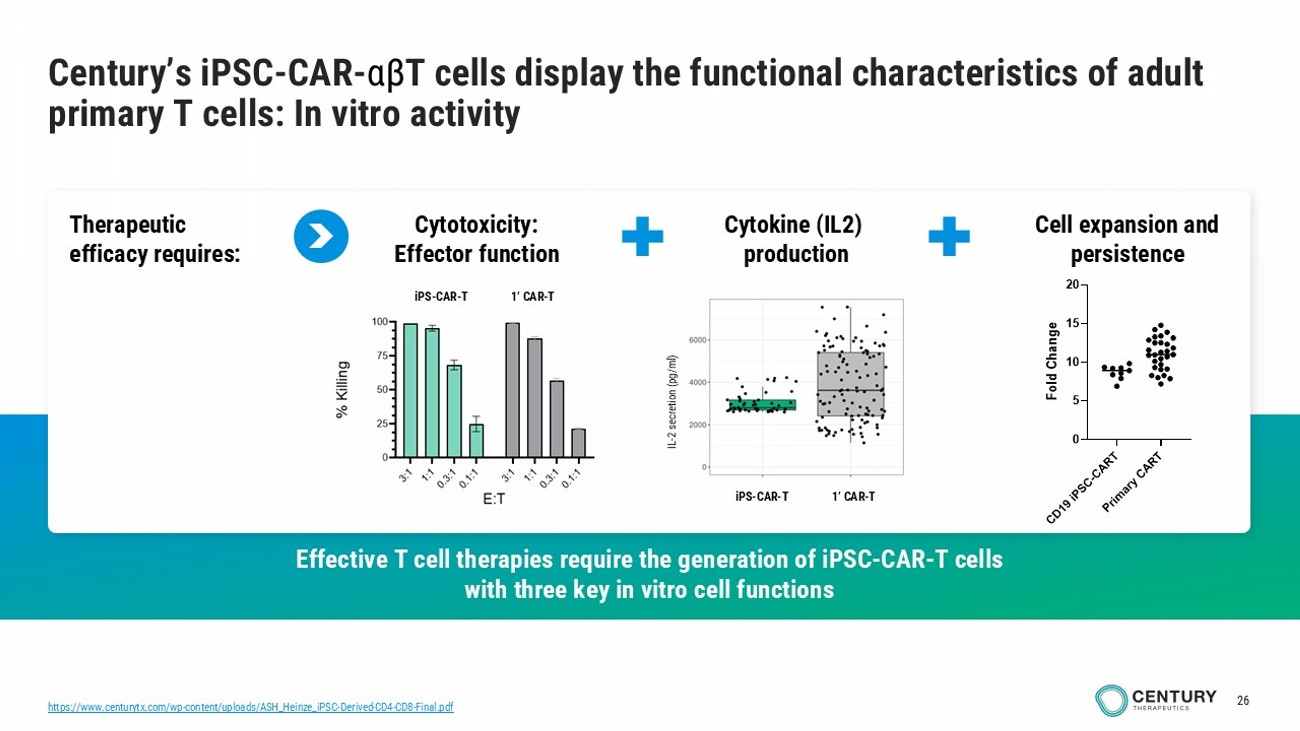
26 Century’s iPSC - CAR - αβ T cells display the functional characteristics of adult primary T cells: In vitro activity Effective T cell therapies require the generation of iPSC - CAR - T cells with three key in vitro cell functions Cytotoxicity: Effector function Cell expansion and persistence Therapeutic efficacy requires: Cytokine (IL2) production iPS - CAR - T 1’ CAR - T iPS - CAR - T 1’ CAR - T IL - 2 secretion (pg/ml) iPS - CAR - T 1’ CAR - T https://www.centurytx.com/wp - content/uploads/ASH_Heinze_iPSC - Derived - CD4 - CD8 - Final.pdf
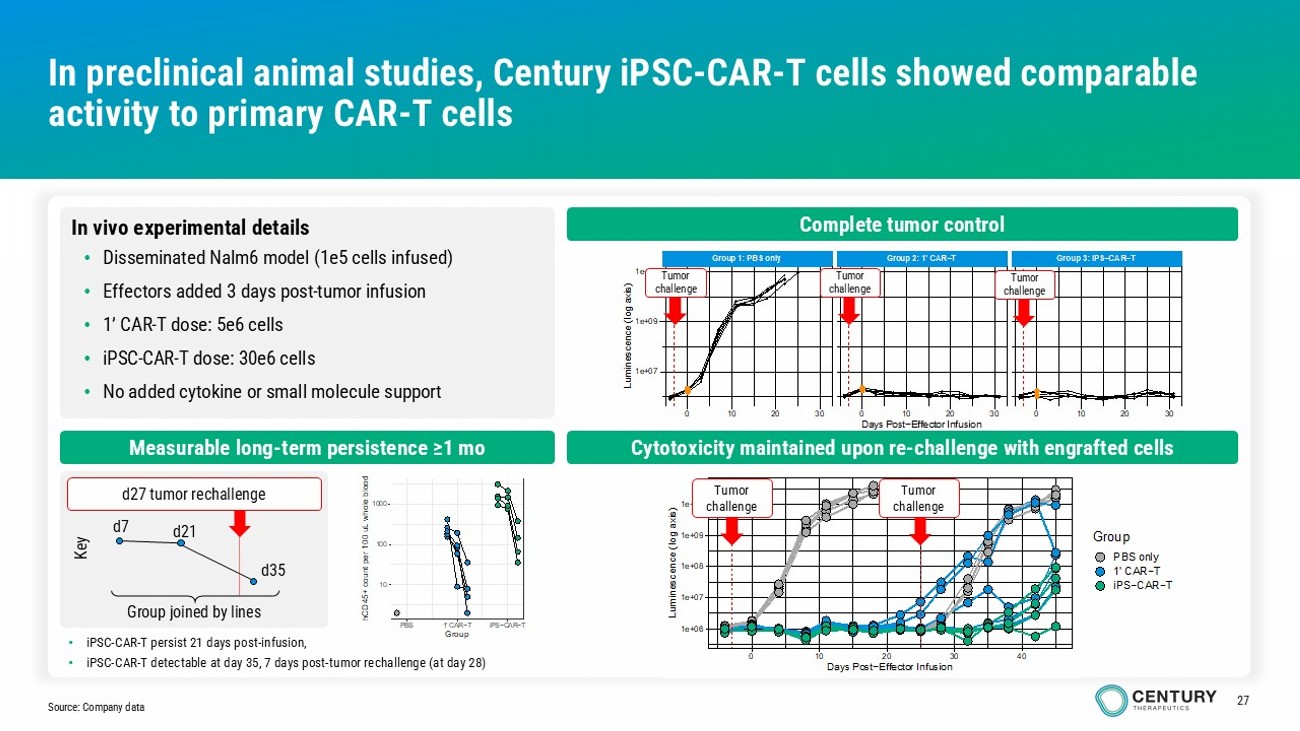
27 1e+06 1e+07 1e+08 1e+09 1e+10 0 10 20 30 40 Days Post−Effector Infusion L u m i n e s c e n c e ( l o g a x i s ) Group PBS only 1' CAR−T iPS−CAR−T Group 1: PBS only Group 2: 1' CAR−T Group 3: iPS−CAR−T 0 10 20 30 0 10 20 30 0 10 20 30 1e+07 1e+09 1e+11 Days Post−Effector Infusion L u m i n e s c e n c e ( l o g a x i s ) In preclinical animal studies, Century iPSC - CAR - T cells showed comparable activity to primary CAR - T cells Source: Company data Complete tumor control Cytotoxicity maintained upon re - challenge with engrafted cells Measurable long - term persistence ≥1 mo 10 100 1000 PBS 1' CAR−T iPS−CAR−T Group h C D 4 5 + c o u n t p e r 1 0 0 u L w h o l e b l o o d In vivo experimental details • Disseminated Nalm6 model (1e5 cells infused) • Effectors added 3 days post - tumor infusion • 1’ CAR - T dose: 5e6 cells • iPSC - CAR - T dose: 30e6 cells • No added cytokine or small molecule support Group joined by lines d7 d21 d35 d27 tumor rechallenge Key Tumor challenge • iPSC - CAR - T persist 21 days post - infusion, • iPSC - CAR - T detectable at day 35, 7 days post - tumor rechallenge (at day 28) Tumor challenge Tumor challenge Tumor challenge Tumor challenge

28 Century’s early - stage programs target best - in - class profiles in oncology 1. Majzner and Mackall. Cancer Discovery. 2018. • αβ CAR - iT cells • Dual - targeting CD19 and CD22 to reduce potential for resistance in malignancy setting • Loss of CD19 in B cell malignancies treated by CD19 CAR T cells is a known mechanism of relapse 1 • Includes Allo - Evasion 5.0 designed to evade host T - cell, NK - cell, and humoral immunity • Incorporates other platform enhancements designed to improve cell persistence and long - term engraftment • CAR iT cells • Novel targeting approaches include • Proprietary nectin - 4 CAR • Novel TCRs providing additional functions • Novel CARs directed to other validated targets • Includes Allo - Evasion 5.0 designed to evade host T - cell, NK - cell, and humoral immunity • Cytokine engineering designed to improve cell persistence post infusion • Engineering designed to enhance trafficking and sustained function in tumor microenvironment CNTY - 341 Solid Tumors

Platform: iPSC cell foundry
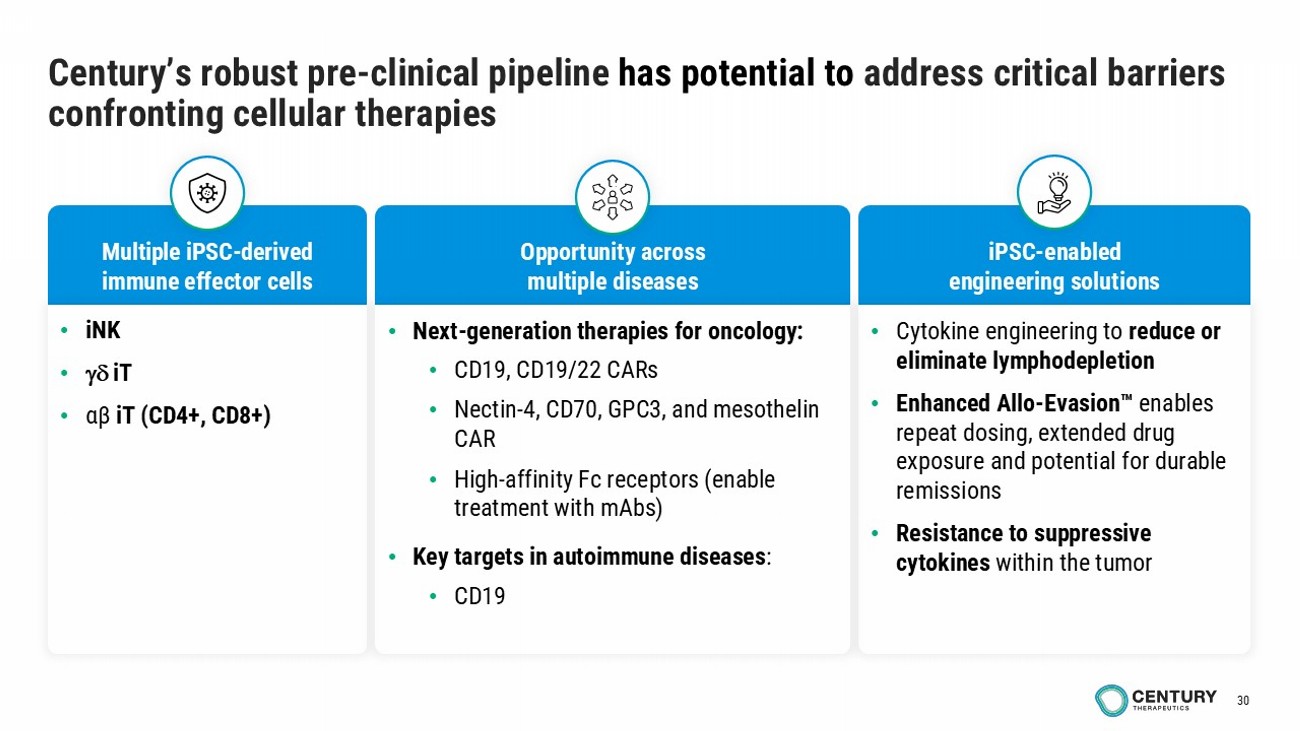
30 Century’s robust pre - clinical pipeline has potential to address critical barriers confronting cellular therapies Multiple iPSC - derived immune effector cells iPSC - enabled engineering solutions Opportunity across multiple diseases • Cytokine engineering to reduce or eliminate lymphodepletion • Enhanced Allo - Evasion enables repeat dosing, extended drug exposure and potential for durable remissions • Resistance to suppressive cytokines within the tumor • iNK • iT • αβ iT (CD4+, CD8+) • Next - generation therapies for oncology: • CD19, CD19/22 CARs • Nectin - 4, CD70, GPC3, and mesothelin CAR • High - affinity Fc receptors (enable treatment with mAbs ) • Key targets in autoimmune diseases : • CD19
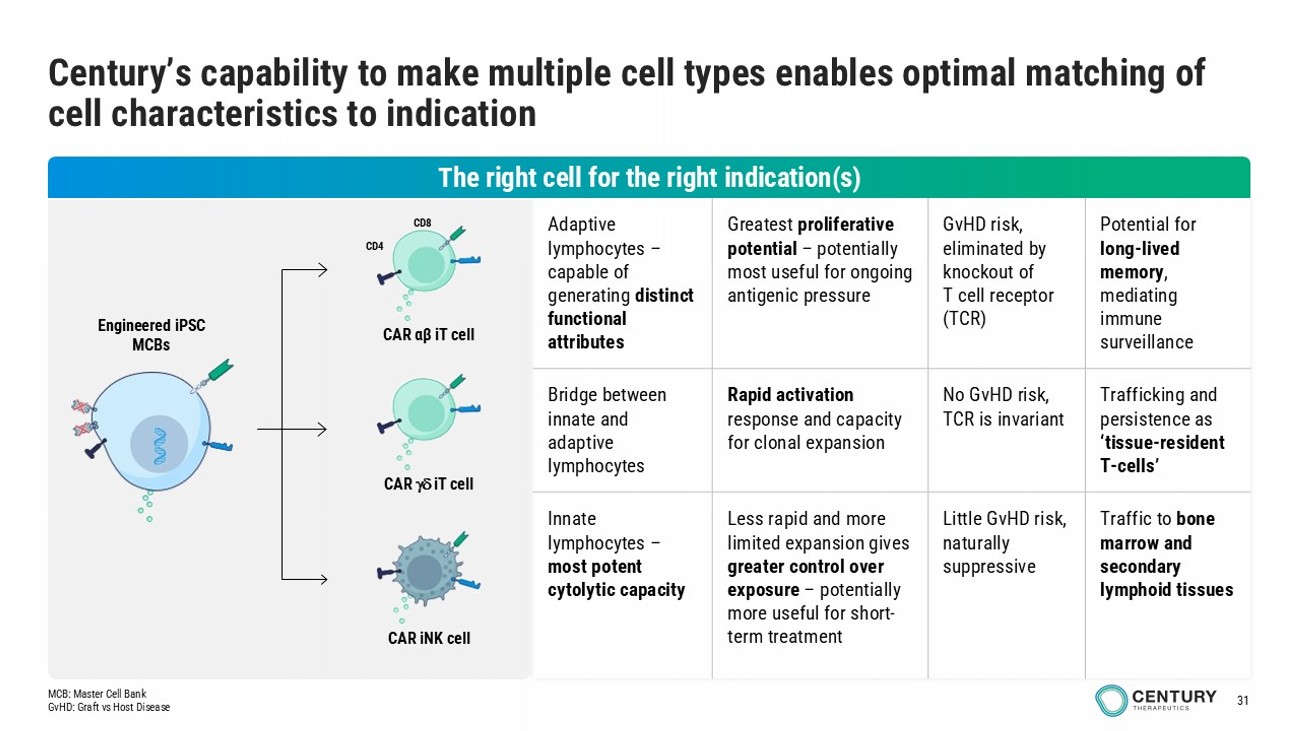
31 Potential for long - lived memory , mediating immune surveillance GvHD risk, eliminated by knockout of T cell receptor (TCR) Greatest proliferative potential – potentially most useful for ongoing antigenic pressure Adaptive lymphocytes – capable of generating distinct functional attributes Trafficking and persistence as ‘tissue - resident T - cells’ No GvHD risk, TCR is invariant Rapid activation response and capacity for clonal expansion Bridge between innate and adaptive lymphocytes Traffic to bone marrow and secondary lymphoid tissues Little GvHD risk, naturally suppressive Less rapid and more limited expansion gives greater control over exposure – potentially more useful for short - term treatment Innate lymphocytes – most potent cytolytic capacity Century’s capability to make multiple cell types enables optimal matching of cell characteristics to indication Engineered iPSC MCBs CAR iNK cell CAR iT cell CAR αβ iT cell CD4 CD8 The right cell for the right indication(s) MCB: Master Cell Bank GvHD: Graft vs Host Disease
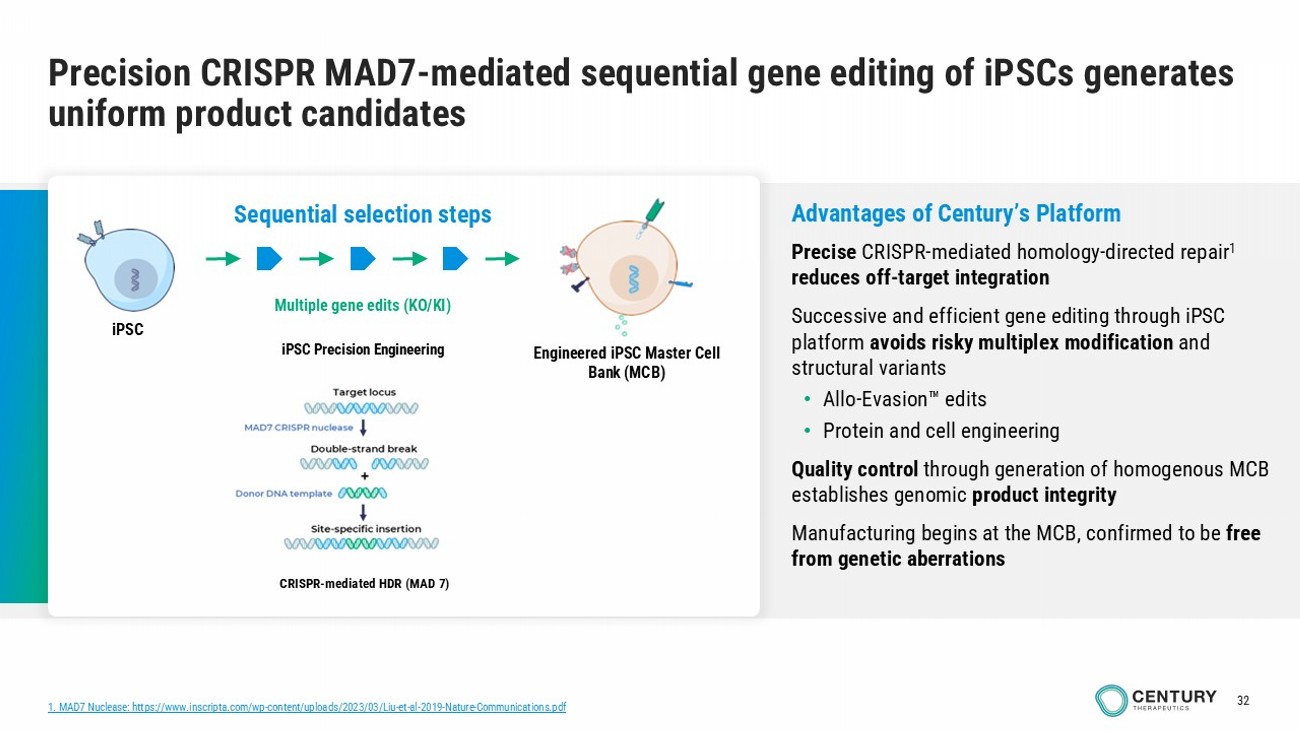
32 Precision CRISPR MAD7 - mediated sequential gene editing of iPSCs generates uniform product candidates Multiple gene edits (KO/KI) iPSC Engineered iPSC Master C ell Bank (MCB) Sequential selection steps iPSC Precision Engineering CRISPR - mediated HDR (MAD 7) Advantages of Century’s Platform Precise CRISPR - mediated homology - directed repair 1 reduces off - target integration Successive and efficient gene editing through iPSC platform avoids risky multiplex modification and structural variants • Allo - Evasion edits • Protein and cell engineering Quality control through generation of homogenous MCB establishes genomic product integrity Manufacturing begins at the MCB, confirmed to be free from genetic aberrations 1. MAD7 Nuclease: https://www.inscripta.com/wp - content/uploads/2023/03/Liu - et - al - 2019 - Nature - Communications.pdf

33 Century platform and in - house manufacturing: Pathway to scalable, profitable cell therapy Quality product at disruptive scale and cost of goods Established in - house manufacturing from development to launch • Consistency : Control of manufacturing and single - donor master - cell - bank over product lifetime for batch - to - batch reproducibility • Increased cell fitness: Differentiated immune cells do not undergo excessive expansion cycles which often result in cell exhaustion • Product homogeneity : Clonal origin enables a well - characterized product • Potential to manufacture at antibody - like scale : Scalable platforms and optimized processes to maximize yield, reduce COGs, and meet demand • Built - for - purpose 53,000 ft 2 cGMP facility • Key leaders each with 1 – 2 decades of cell therapy manufacturing expertise, from leading commercial cell therapies • In - house team facilitates aligned priorities, learnings, faster product iteration for efficiency, speed, and product quality • Builds and protects proprietary know - how • Optionality with redundant sites (in - house, active CDMO)
Extending cash runway into 4Q26; re - allocating resources to enable key value milestones Enhanced preclinical pipeline and platform aiming to expand and multiply cell therapy value • Four potentially transformative programs engineered with industry - leading Allo - Evasion 5.0 • Leading programs focus on iPSC - derived ‘tunable’ CD4+/CD8+ αβ T cells • Selective expansion to non - immune effector cells in high impact diseases Enhanced Preclinical Pipeline Concentrated Clinical Focus Resourcing for Value Concentrating clinical focus with CNTY - 101 on autoimmune disorders with transformational potential • Unique profile of CD19 - targeting iNK cell product engineered with Allo - Evasion with clinical data from R/R NHL reinforcing potential in autoimmune disorders • Expansion of Phase 1 CALiPSO - 1 trial in US and EU with CARAMEL IIT expected to commence in mid - 2025 Century Therapeutics: Clear focus on transformational value with unique iPSC - derived cell therapies GOAL: E xpand and enhance cell therapy value • Ended FY24 with cash, cash equivalents, and investments of $220M • CNTY - 308 T cell program expected to enter IND - enabling stage in mid - 2025 • CNTY - 101 autoimmune clinical data expected in 2025
www.centurytx.com
